

Afonso Caricati-Neto*, Leandro Bueno Bergantin
Department of Pharmacology, Laboratory of Autonomic and Cardiovascular Pharmacology, Escola Paulista de Medicina, Universidade Federal de São Paulo (UNIFESP), 55 11 5576-4973, Rua Pedro de Toledo, 669 - Vila Clementino, São Paulo - SP, Brazil, Postal Code: 04039-032.
*Corresponding Author
Prof. Dr. Afonso Caricati-Neto
Department of Pharmacology,
Laboratory of Autonomic and Cardiovascular Pharmacology,
Escola Paulista de Medicina, Universidade Federal de São Paulo (UNIFESP), 55 11 5576-4973, Rua Pedro de Toledo, 669 - Vila Clementino, São Paulo - SP, Brazil, Postal Code: 04039-032.
Abstract
It is well established that an imbalance of neuronal Ca2+ homeostasis contributes to the pathogenesis of neurodegenerative diseases, such as Alzheimer's (AD) and Parkinson's (PD) diseases. Therefore, regulation of neuronal Ca2+ homeostasis may represent a new therapeutic strategy of these diseases. Our recent discovery of the involvement of the interaction between intracellular signaling pathways mediated by Ca2+ and cAMP (Ca2+/cAMP signaling interaction) in the neurotransmission and neuroprotection, and its pharmacological modulation, has contributed to the understanding of pathophysiology and pharmacology of neurodegenerative diseases. This discovery emerged from numerous clinical studies performed since 1975 that reported that the use of L-type Ca2+ channel blockers (CCBs) during the antihypertensive therapy decreased arterial pressure, but produced typical symptoms of sympathetic hyperactivity such as tachycardia and increment of catecholamine plasma levels. Despite these adverse effects have been initially attributed to adjust reflex of arterial pressure, during almost four decades this phenomenon remained unclear. In 2013, we discovered that this sympathetic hyperactivity is due to increase of transmitter release from sympathetic neurons and adrenal chromaffin cells caused by the modulatory action of CCBs on the Ca2+/cAMP signaling interaction. Additionally, we discovered that the pharmacological modulation of neural Ca2+/cAMP signaling interaction can reduce neuronal death due to attenuation of cytosolic Ca2+ overload and stimulation of cell survival pathways mediated by cAMP-response element binding protein (CREB). Then, our discovery of the pharmacological modulation of neural Ca2+/cAMP signaling interaction may open a large avenue for the development of a new therapeutic strategy for AD and PD.
Keywords: Ca2+/cAMP signaling interaction; Alzheimer’s disease, Parkinson’s disease, neurological/psychiatric disorders
Introduction
The growing increase in the life expectancy of the world's population has increased the concern about neurodegenerative diseases related to aging, such as Alzheimer's (AD) and Parkinson’s (PD) diseases. According to a 2015 United Nations report on world population ageing, the number of people aged 60 and older worldwide is projected to more than double in the next 35 years, reaching almost 2.1 billion people. Most of this growth will come from developing regions of the world, although the oldest old, who are more than 80 years of age, are the fastest growing segment of the population in developed regions. Despite these improvements in life expectancy, AD and PD, and related neurodegenerative conditions have arguably become the most dreaded maladies of older people.
It is well established that an imbalance of neuronal Ca2+ homeostasis contributes to the pathogenesis of neurodegenerative diseases, including AD and PD. Therefore, regulation of neuronal Ca2+ homeostasis may represent a new therapeutic strategy of these diseases. Recent studies showed that the treatment with L-type CCBs reduces motor symptoms and attenuates progressive neuronal death in animal model of degenerative disease (Ilijic et al., 2011; Wu et al., 2016), suggesting that the blockade of Ca2+ influx produced by L-type CCBs could attenuate or prevent neuronal death in neurodegenerative diseases, such as PD and AD. However, the mechanisms involved in these effects of L-type CCBs remains unclear.
Since 1970´s, several clinical studies have reported that acute and chronic administration of L-type Ca2+ channel blockers (CCBs) during antihypertensive therapy, such as nifedipine and verapamil, decreased arterial pressure but produced typical symptoms of sympathetic hyperactivity, such as tachycardia and enhance of catecholamine plasma levels (Grossman and Messerli, 1998). Despite of these effects have been initially attributed to adjust reflex of arterial pressure, the mechanisms involved in these adverse effects remained unclear for decades. Some studies performed since 1975 in isolated tissues richly innervated by sympathetic nerves (rodent vas deferens), to exclude the influence of adjusting reflex, showed that responses produced by neurotransmitter released from these nerves were completely inhibited by L-type CCBs in high concentrations (>1 μmol/L), but paradoxically potentiated in concentrations below 1 μmol/L (Kreye et al., 1975; French et al., 1981; Moritoki et al., 1987; Bergantin et al., 2013). The molecular mechanisms responsible to this paradoxical sympathetic hyperactivity induced by CCBs, defined by us as “calcium paradox”, remained unclear for almost forty years.
In 2013, we discovered that this paradoxical sympathetic hyperactivity resulted from the increase of transmitter release from sympathetic neurons and adrenal chromaffin cells stimulated by CCBs due to its modulatory action on the interaction between the intracellular signaling pathways mediated by Ca2+ and cAMP (Ca2+/cAMP signaling interaction) in these cells (Bergantin et al., 2013). In addition, we discovered that this modulatory action of CCBs enhances the intracellular cAMP levels, which in turn reduce neuronal death resulting from cytosolic Ca2+ overload due probably to activation of cellular survival pathways mediated by cAMP-response element binding protein (CREB) (Caricati-Neto et al., 2015; Bergantin et al., 2015; Bergantin and Caricati-Neto, 2016a, 2016b, 2016c, 2016d). Then, our discovery of the role of the Ca2+/cAMP signaling interaction in neurotransmission and neuroprotection, and its pharmacological modulation, may open a large avenue for the development of a new therapeutic strategy for neurological/psychiatric disorders.
Our studies have proposed that the pharmacological modulation of this signaling interaction by use of the L-type CCBs and compounds that increase the cytosolic concentration of cAMP (cAMP-enhancer compounds) could be effective in enhancing neurotransmission and neuroprotection in neurological/psychiatric disorders resulting from neurotransmission deficit and neuronal death, such as such as AD and PD (Bergantin et al., 2013; Caricati-Neto et al., 2015; Bergantin et al., 2015; Bergantin and Caricati-Neto, 2016a, 2016b, 2016c, 2016d). Then, in this review we will discuss how the pharmacological modulation of the Ca2+/cAMP signaling interaction could be a new therapeutic strategy to treat the neurodegenerative diseases, especially AD and PD.
Role of the neural Ca2+/cAMP signaling interaction in neurotransmission
Experimental studies initiated decades ago, using adrenal chromaffin cells as cellular model, established the notion of stimulus-secretion coupling to explain transmitter release from central and peripheral neurons. In 1970´s was discovered that an increase of the cytosolic Ca2+ concentration ([Ca2+]c) constitutes a basic requirement to trigger release by exocytosis of secretory vesicles containing transmitter (catecholamines, purines and other substances) in adrenal chromaffin cells (Baker et al., 1978). Later, it was discovered a direct relationship between rise in [Ca2+]c and rapid transmitter release from chromaffin cells (Neher et al., 1993). In addition to Ca2+, other intracellular messengers are involved in the exocytosis of neurotransmitter and hormones. In 1970´s was discovered that elevation of intracellular cAMP concentration ([cAMP]c) mediated by activation adenylyl cyclases (ACs) with forskolin enhanced exocytosis of secretory vesicles in adrenal chromaffin cells (Chern et al., 1988), suggesting that both Ca2+ and cAMP participate of transmitter exocytosis.
It is well established that the blockade of Ca2+ influx through the L-type voltage-activated Ca2+ channels (VACC) reduce transmitter exocytosis from chromaffin cells and neurons. But, using isolated tissues richly innervated by sympathetic nerves (rat vas deferens) stimulated by electrical pulses, we showed that responses mediated by these nerves were reduced and completely inhibited by L-type CCBs in high concentrations (>1 μmol/L) due to blockade of L-type VACC, but paradoxically increased in concentrations below 1 μmol/L (Bergantin et al., 2013). Although this CCBs-induced sympathetic hyperactivity, defined by us as “calcium paradox”, had been previously reported by several studies (Kreye et al., 1975; French et al., 1981; Moritoki et al., 1987), the molecular mechanisms responsible for this adverse effect remained unclear.
In 2013, we discovered that these CCBs-induced sympathetic hyperactivity was potentiated by cAMP-enhancer compounds, such as ACs activators (forskolin) and phosphodiesterase (PDE) inhibitors (rolipram and isobutyl methyl xanthine (IBMX)) (Bergantin et al., 2013). This potentiation by cAMP-enhancer compounds was prevented by inhibition of ACs with SQ 22536 (Bergantin et al., 2013). As the activity of ACs is regulated by Ca2+, the reduction of [Ca2+]c produced by L-type CCBs in high concentrations results in increase of activity of ACs and elevation of [cAMP]c (Bergantin et al., 2013). The elevation of [cAMP]c activates cyclic AMP-dependent protein kinase or A kinase (PKA) that activate endoplasmic reticulum (ER) Ca2+ channels, such as ER-Ca2+ channels regulated by ryanodine receptors (RyR), stimulating Ca2+ release (Bergantin et al., 2013). This PKA-stimulated Ca2+ release from ER enhances number of secretory vesicles docked in plasma membrane, increasing neurotransmitter release and synaptic concentration of neurotransmitters (Bergantin et al., 2013). These findings led to the discovery that reduction of Ca2+ influx through L-type VACC produced by CCBs stimulates ACs/cAMP/PKA signaling pathway, increasing neurotransmitter release and consequently synaptic transmission due to interaction between intracellular signaling pathways mediated by Ca2+ and cAMP, defined by us as Ca2+/cAMP signaling interaction (Bergantin et al., 2013; Caricati-Neto et al., 2015; Bergantin and Caricati-Neto, 2016a, 2016b, 2016c, 2016d).
Our discovery solved the enigmatic “calcium paradox” of almost four decades involved in sympathetic hyperactivity produced by L-type CCBs due to its modulatory action on the neural Ca2+/cAMP signaling interaction (Bergantin et al., 2013; Caricati-Neto et al., 2015; Bergantin and Caricati-Neto, 2016a, 2016b, 2016c, 2016d). This discovery strengthened the idea that the exocytosis of secretory vesicles containing transmitters from neurons and neuroendocrine cells is finely regulated by neural Ca2+/cAMP signaling interaction (Bergantin et al., 2013; Caricati-Neto et al., 2015; Bergantin and Caricati-Neto, 2016a, 2016b, 2016c, 2016d). Figure 1 shows the potentiation by cAMP-enhancer compounds of the CCBs-induced sympathetic hyperactivity.
Figure 1. Increase of neurotransmission produced by pharmacological modulation of neural Ca2+/cAMP signaling interaction. (A) Records showing that contractile responses mediated by neurotransmitter released from sympathetic nerves by means electrical field stimulation in rat vas deferens (neurogenic contractions) were significantly reduced by L-type CCBs (verapamil) in high concentrations (>10-6 M), but paradoxically increased in concentrations below 10-6 M, characterizing CCBs-induced sympathetic hyperactivity. This increase of neurogenic contractions by verapamil (<10-6 M) was potentiated by pre-treatment of isolated tissue with cAMP-enhancer compounds, such as rolipram 10-7 M (B), IBMX 10-6 M (C) and forskolin 10-7 M (D). This potentiation by cAMP-enhancer compounds was prevented by inhibition of ACs with SQ 22536 (Data not showed). Each point below the record represents molar concentration of verapamil (interval of 0.5 log unity). Each line below the record represents incubation time with cAMP-enhancer compounds. Representative records extracted from Bergantin et al. (2013).
Our studies showed that the pharmacological modulation of neural Ca2+/cAMP signaling interaction by combined use of the L-type CCBs and cAMP-enhancer compounds may enhance neurotransmitter release causing increase of synaptic transmission (Bergantin et al., 2013; Caricati-Neto et al., 2015; Bergantin and Caricati-Neto, 2016a, 2016b, 2016c, 2016d). Then, the pharmacological modulation of this interaction could be a new strategy to increase neurotransmission in neurodegenerative disease characterized by severe deficit in central neurotransmission, such as AD and PD.
Role of the neural Ca2+/cAMP signaling interaction in neuroprotection
It is well established that an imbalance of neuronal Ca2+ homeostasis, especially cytosolic Ca2+ overload, contributes to the pathogenesis of neurodegenerative diseases, such as AD and PD (Bergantin et al., 2013; Caricati-Neto et al., 2015; Bergantin et al., 2015; Bergantin and Caricati-Neto, 2016a, 2016b, 2016c, 2016d). Therefore, regulation of neuronal Ca2+ homeostasis may represent a new strategy for treatment of these diseases. As previously mentioned, blockade of the L-type VACC by CCBs reduces Ca2+ influx and [Ca2+]c, but produces increase of [cAMP]c (Bergantin et al., 2013; Caricati-Neto et al., 2015; Bergantin and Caricati-Neto, 2016a, 2016b, 2016c, 2016d). This functional Ca2+/cAMP signaling interaction regulates various cellular responses, including neurotransmitter release (Bergantin et al., 2013; Caricati-Neto et al., 2015; Bergantin and Caricati-Neto, 2016a, 2016b, 2016c, 2016d).
Recent studies showed that the treatment with L-type CCBs reduced motor symptoms and attenuated progressive neuronal death in animal model of degenerative disease, suggesting that L-type CCBs are potentially viable neuroprotective agents (Ilijic et al., 2011; Wu et al., 2016). These studies reinforced the idea that attenuation of cytosolic Ca2+ overload produced by L-type CCBs due to blockade of Ca2+ influx through L-type VACC could be an excellent pharmacological strategy to attenuate or prevent neuronal death in neurodegenerative diseases.
Some studies showed that increase of [cAMP]c may stimulate neuroprotective response and attenuate neuronal death due to activation of cellular survival pathways mediated by cAMP/PKA/ cAMP-response element binding protein (CREB)-dependent intracellular signaling pathway (Sommer et al., 1995; Onozuka et al., 2008; Xiao et al., 2011; Li et al., 2016). Then, the pharmacological modulation of the neural Ca2+/cAMP signaling interaction by combined use of L-type CCBs and cAMP-enhancer compounds could stimulate neuroprotective response due to dual action mediated by increase of [cAMP]c and attenuation of cytosolic Ca2+ overload (Bergantin et al., 2013; Caricati-Neto et al., 2015; Bergantin and Caricati-Neto, 2016a, 2016b, 2016c, 2016d). Our studies suggest that pharmacological modulation of this interaction could be a new neuroprotective therapeutic strategy to slow the progression of neurodegenerative diseases, such as AD and PD. Figure 2 shows how pharmacological modulation of the neural Ca2+/cAMP signaling interaction by combined use of L-type CCBs and cAMP-enhancer compounds can produce increase of neurotransmission and neuroprotection.
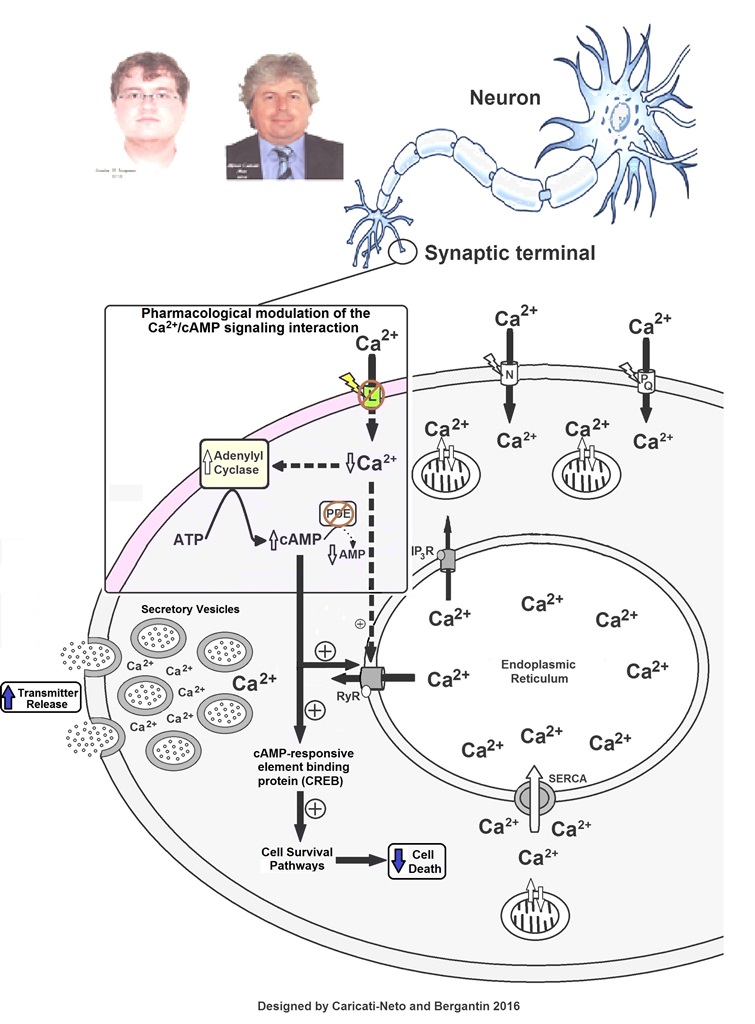
Figure 2. Increase of neurotransmitter release and attenuation of neuronal death (neuroprotection) produced by pharmacological modulation of the neural Ca2+/cAMP signaling interaction by combined use of L-type Ca2+ channel blockers (CCBs) and cAMP-enhancer drugs. PDE - phosphodiesterase inhibitors. Figure extracted from Bergantin and Caricati-Neto, 2016d).
Pharmacological modulation of neural Ca2+/cAMP signaling interaction as a new therapeutic strategy of Alzheimer’s disease (AD)
AD is a progressive neurodegenerative disorder related to ageing characterized by cognitive and memory deterioration. Neuritic plaques represent the pathological status of AD, and are respectively related to the accumulation of the β-amyloid peptide (Aβ) in brain tissues (De-Paula et al., 2012; Mohamed et al., 2016). According to the amyloid hypothesis, the overproduction of Aβ is a consequence of the disruption of homeostatic processes that regulate the proteolytic cleavage of the amyloid precursor protein (APP). Genetic and age-related factors could contribute to a metabolic change, favoring the amyloidogenic processing of APP in detriment of the physiological secretory pathway (De-Paula et al., 2012; Mohamed et al., 2016).
The neurotoxic potential of the Aβ results from its biochemical properties that favor aggregation. These processes, along with a reduction of Aβ clearance from the brain, leads to the extracellular accumulation of Aβ, and the subsequent activation of neurotoxic cascades that ultimately lead to cytoskeletal changes, neuronal dysfunction and cellular death (De-Paula et al., 2012). Intracerebral amyloidosis development in AD patients is in an age-dependent manner, but recent evidences indicate that it may be observed in some subjects as early as in the third or fourth decades of life, with increasing magnitude in late middle age, and highest estimates in old age (De-Paula et al., 2012; Mohamed et al., 2016; Zamponi, 2016).
Therapies targeting the modification of amyloid-related cascades may be viewed as promising strategies to attenuate or even to prevent dementia (De-Paula et al., 2012). Therefore, the cumulative knowledge on the pathogenesis of AD derived from basic science models will hopefully be translated into clinical practice in the forthcoming years. Other targets relevant to AD have also been considered in the last years for producing multitarget compounds (Maroto et al., 2011; Ismaili et al., 2016;).
In addition to what has been discussed above, acetylcholinesterase (AChE) is another important target to treat the pathogenesis of AD (cholinergic dysfunction hypothesis). Considering the current hypothesis of accumulation of the Aβ in AD, this relies in the reduction of neurotransmitter acetylcholine (ACh) release in central cholinergic nervous system involved in cognitive function. Thus, the inhibition of ACh degradation by AChE is a potential target to treat AD (Ismaili et al., 2016; Maroto et al., 2011; Samochocki et al., 2003, Caricati-Neto et al., 2004).
An imbalance of intracellular Ca2+ homeostasis also contributes to the pathogenesis of neurodegenerative diseases, including AD. Several evidences suggest that aging impairs ability of the brain intracellular Ca2+ degradation which is likely to induce cellular damage due to cytosolic Ca2+ overload leading to neural death and resultant cognitive dysfunction, such as AD (Kawamoto et al., 2012). Therefore, regulation of intracellular Ca2+ homeostasis may represent a new therapeutic strategy of AD.
A 10-year follow-up study (2000 to 2010) involving 82,107 hypertensive patients of more than 60 years of age, showed that use of L-type CCBs reduced blood pressure and risk of dementia in hypertensives, suggesting that these drugs could be clinically used to treat AD (Wu et al., 2016). Supportive findings for the neuroprotective effects of CCBs have been demonstrated in 1,241 elderly hypertensive patients with memory impairment (Hanon et al., 2006). The use of CCBs decreased the risk of cognitive impairment and AD independently of BP levels when compared to patients not receiving CCBs (Hanon et al., 2006). The long-term effects of antihypertensive therapy initiated with a long-acting dihydropinidine (nitrendipine) has been demonstrated in the double-blind, placebo-controlled Syst-Eur trail in which the incidence of dementia was reduced by 55 % (Forette et al., 2002).
Some studies have proposed that hybrid compounds having the moieties of tacrine, a potent inhibitor of brain and peripheral AChE, and nimodipine, a L-type CCBs could be useful to treatment of AD (Maroto et al., 2011; Ismaili et al., 2016). In addition, galantamine, a mild AChE inhibitor and an allosteric ligand of nicotinic receptors has been used to improve cognition and behaviour in patients with AD (Samochocki et al., 2003; Caricati-Neto et al., 2004).
It was showed in AD model rats that cAMP-enhancer compounds, such as nobiletin (a polymethoxylated flavone from citrus peels) and oxyntomodulin (a proglucagon-derived peptide that co-activates the GLP-1 receptor and the glucagon receptor), produce neuroprotective effect mediated by intracellular cAMP production, activation of PKA and MAPK pathways and phosphorylation of CREB (Onozuka et al., 2008; Li et al., 2016).
Our discovery of the involvement of the Ca2+/cAMP signaling interaction in the neurotransmission and neuroprotection has produced important advances in the understanding of the pathophysiology and pharmacology of AD (Bergantin et al., 2013; Caricati-Neto et al., 2015; Bergantin et al., 2015; Bergantin and Caricati-Neto, 2016a, 2016b, 2016c, 2016d). These advances allowed us to propose that pharmacological modulation of the Ca2+/cAMP signaling interaction produced by combined use of the L-type CCBs (used in the antihypertensive therapy), such as isradipine, and cAMP-enhancer compounds (used in the anti-depressive therapy), such as rolipram, could represent a new therapeutic strategy of AD in humans.
Pharmacological modulation of the Ca2+/cAMP signaling interaction by combined use of the L-type CCBs and cAMP-enhancer drugs could attenuate ACh release deficit, increasing central cholinergic neurotransmission involved in the control of cognitive function. In addition, pharmacological modulates of this interaction could contribute to reduce neuronal death due to attenuation of cytosolic Ca2+ overload, increase of [cAMP]c and stimulation of cell survival pathways probably mediated by activation of cellular survival pathways regulated by AMP/PKA/CREB-dependent intracellular signaling pathway (Sommer et al., 1995; Onozuka et al., 2008; Xiao et al., 2011; Li et al., 2016). Thus, pharmacological modulation of Ca2+/cAMP signaling interaction could be a new neuroprotective therapeutic strategy to slow the progression of AD (Bergantin et al., 2013; Caricati-Neto et al., 2015; Bergantin et al., 2015; Bergantin and Caricati-Neto, 2016a, 2016b, 2016c, 2016d).
Pharmacological modulation of neural Ca2+/cAMP signaling interaction as a new therapeutic strategy of Parkinson’s disease (PD)
PD is an age-associated progressive neurodegenerative disorder that has gained crescent social and economic impact due to the aging of the western society. While the distinctive motor symptoms of PD have been described for centuries, cognitive impairment has only recently been recognized as a central feature. All current therapies are symptomatic and fail to reverse or halt the progression of dopaminergic neurons loss.
Dopamine loss in the substantia nigra, which results from reduction of dopamine release in striatal dopaminergic neurons due to neuronal death, outcomes in the recognizable core signs of asymmetrical bradykinesia and hypokinesia (slowness and reduced amplitude of movement), muscle rigidity (stiffness) and rest tremor, consequences from modifying motor control. Rest tremor, prominent asymmetry and a good response to levodopa are the features that most accurately predict PD pathology (Hayes et al., 2010; Salat et al., 2016). The tremor-dominant form of PD tends to run a more benign course than typical PD. Early falls or autonomic symptoms, and a response to PD medicines should raise evidences about the diagnosis (Hayes et al., 2010; Salat et al., 2016). Medication-induced parkinsonism due to commonly prescribed dopamine-blocking medications, such as antipsychotics (eg, haloperidol, risperidone) and antiemetics (eg, metoclopramide, prochlorperazine) should be excluded in PD patients.
Functional imaging of the dopaminergic system using cerebral single photon emission computed tomography or positron emission tomography can be useful in diagnosis of early Parkinson. Positron emission tomography studies examining the rate of decline in dopamine-producing cells suggest that humans have already lost 50%–70% of their nigral neurons, before they develop motor symptoms, and it has been estimated that the duration of this “presymptomatic” phase is about 5 years (Hayes et al., 2010; Salat et al., 2016).
Early diagnoses will become a critical issue if effective neuroprotective drugs become available. In fact, increasing dopamine, mainly by levodopa combined with a dopa-decarboxylase inhibitor remains the most potent drug therapy for reversing motor impairment. A higher maintenance dose of levodopa (eg, 200 mg three times daily compared with an initial dose of 100 mg three times daily) provides slightly greater benefit for reducing motor symptoms, but at the cost of earlier wearing-off symptoms and dyskinesias (Hayes et al., 2010; Salat et al., 2016).
The combination of novel concepts may lead to advances in PD research with the promise of finding compounds that are both effective, and fast-acting, including in patients who have tried other therapies with limited success. In conclusion, new insights for more efficient pharmacological treatments of PD are clearly needed.
Several evidences suggest that an imbalance of intracellular Ca2+ homeostasis contributes to the pathogenesis of PD, thus regulation of intracellular Ca2+ homeostasis may represent a new strategy for treatment of PD. Several evidences suggest that aging impairs ability of the brain intracellular Ca2+ degradation which is likely to induce cellular damage leading to death of dopaminergic neurons in the substantia nigra and resultant motor dysfunction due to reduction of dopamine release in striatal dopaminergic neurons.
A phase II clinical trial published in 2016 showed that treatment with isradipine was safely tolerated to reduce motor symptoms by PD patients (Swart et al., 2016). In addition, it was showed that the treatment with L-type CCBs such as isradipine reduces motor symptoms and attenuates progressive death of dopamine neurons from substantia nigra in animal model of PD (Ilijic et al., 2011). This study showed that isradipine produces a dose-dependent sparing of dopaminergic fibers and cell bodies at concentrations achievable in humans (Ilijic et al., 2011), suggesting that L-type CCBs are potentially viable neuroprotective agents useful for PD treatment. These finding reinforced the idea that attenuation of cytosolic Ca2+ overload produced by L-type CCBs due to blockade of Ca2+ influx through L-type VACC could be an excellent pharmacological strategy to attenuate or prevent neuronal death in PD.
Our discovery of the involvement of the Ca2+/cAMP signaling interaction in the neurotransmission and neuroprotection has contributed to understanding of the pathophysiology and pharmacology of PD (Bergantin et al., 2013; Caricati-Neto et al., 2015; Bergantin et al., 2015; Bergantin and Caricati-Neto, 2016a, 2016b, 2016c, 2016d). These advances allowed us to propose that pharmacological modulation of the Ca2+/cAMP signaling interaction produced by combined use of the L-type CCBs and cAMP-enhancer compounds could represent a new therapeutic strategy of PD in humans.
Pharmacological modulation of the Ca2+/cAMP signaling interaction could attenuate dopamine release deficit, increasing central dopaminergic neurotransmission involved in the control of motor function. In addition, pharmacological modulates of this interaction could contribute to reduce death of dopaminergic neurons due to attenuation of cytosolic Ca2+ overload, increase of [cAMP]c and stimulation of cell survival pathways probably mediated by activation of cellular survival pathways regulated by AMP/PKA/CREB-dependent intracellular signaling pathway (Sommer et al., 1995; Onozuka et al., 2008; Xiao et al., 2011; Li et al., 2016). Thus, pharmacological modulation of Ca2+/cAMP signaling interaction could be a new neuroprotective therapeutic strategy to slow the progression of PD (Bergantin et al., 2013; Caricati-Neto et al., 2015; Bergantin et al., 2015; Bergantin and Caricati-Neto, 2016a, 2016b, 2016c, 2016d).
Conclusion
Our recent discovery of the Ca2+/cAMP signaling interaction could promote important advances in the pathophysiology and pharmacology of the neurological/psychiatric disorders. These advances can contribute to drug development more effective and safer to prevent clinical symptoms of AD and PD.
Disclosure Statement
Caricati-Neto and Bergantin thank the continued financial support from CAPES, CNPq and FAPESP (Bergantin´s Postdoctoral Fellowship FAPESP #2014/10274-3).
The authors also thank Elsevier - “author use”: Reuse of portions or extracts from the article in other works -
https://www.elsevier.com/__data/assets/pdf_file/0007/55654/AuthorUserRights.pdf.
References
Baker PF, Knight DE. 1978. Calcium-dependent exocytosis in bovine adrenal medullary cells with leaky plasma membranes. Nature 276: 620–622.
Bergantin LB, Souza CF, Ferreira RM, Smaili SS, Jurkiewicz NH, Caricati-Neto A, Jurkiewicz A. 2013. Novel model for “calcium paradox” in sympathetic transmission of smooth muscles: role of cyclic AMP pathway. Cell Calcium 54: 202–212.
Bergantin LB, Jurkiewicz A, García AG, Caricati-Neto A. 2015. A Calcium Paradox in the Context of Neurotransmission. Journal of Pharmacy and Pharmacology 3: 253-261.
Bergantin LB, Caricati-Neto, A. 2016a. Challenges for the pharmacological treatment of neurological and psychiatric disorders: Implications of the Ca2+/cAMP intracellular signalling interaction. European Journal Pharmacology 788, 255-260.
Bergantin LB, Caricati-Neto A. 2016b. Insight from “Calcium Paradox” due to Ca2+/cAMP Interaction: Novel Pharmacological Strategies for the Treatment of Depression. International Archives of Clinical Pharmacology 2: 007.
Bergantin LB, Caricati-Neto A. 2016c. Novel Insights for Therapy of Parkinson’s disease: Pharmacological Modulation of the Ca2+/cAMP Signalling Interaction. Austin Neurology & Neurosciences 1(2): 1009.
Bergantin LB, Caricati-Neto A. 2016d. Recent advances in pharmacotherapy of neurological and psychiatric disorders promoted by discovery of the role of Ca2+/cAMP signaling interaction in the neurotransmission and neuroprotection. Advance Pharmaceutical Journal 1(3): 66.
Caricati-Neto A, D'angelo LC, Reuter H, Hyppolito Jurkiewicz N, Garcia AG, Jurkiewicz A. 2004. Enhancement of purinergic neurotransmission by galantamine and other acetylcholinesterase inhibitors in the rat vas deferens. European Journal Pharmacology 503(1-3): 191-201.
Caricati-Neto A, García AG, Bergantin LB. 2015. Pharmacological implications of the Ca2+/cAMP signalling interaction: from risk for antihypertensive therapy to potential beneficial for neurological and psychiatric disorders. Pharmacology Research and Perspectives 3(5): e00181.
Chern YJ, Kim KT, Slakey LL, Westhead EW. 1988. Adenosine receptors activate adenylate cyclase and enhance secretion from bovine adrenal chromaffin cells in the presence of forskolin. Journal of Neurochemistry 50: 1484–1493.
De-Paula VJ, Radanovic M, Diniz BS, Forlenza OV. 2012. Alzheimer's disease. Subcellular Biochemistry, 65:329-52.
Forette F, Seux ML, Staessen JA, Thijs L, Babarskiene MR, Babeanu S et al. 2002. Systolic Hypertension in Europe Investigators The prevention of dementia with antihypertensive treatment: new evidence from the systolic hypertension in Europe (Syst-Eur) study. Archives of Internal Medicine 162(18): 2046–2052.
French AM, Scott NC. 1981. A comparison of the effects of nifedipine and verapamil on rat vas deferens. British Journal Pharmacology 73: 321–323.
Grossman E, Messerli FH. 1998. Effect of calcium antagonists on sympathetic activity. European Heart Journal 19 (Suppl F): F27–F31.
Hanon O, Pequignot R, Seux ML, Lenoir H et al. 2006. Relationship between antihypertensive drug therapy and cognitive function in elderly hypertensive patients with memory complaints. Journal of Hypertension 24(10): 2101–2107.
Hayes MW, Fung VS, Kimber TE, O'Sullivan JD. 2010. Current concepts in the management of Parkinson disease. Medicine Journal Australia 192, 144-149.
Ilijic E, Guzman JN, Surmeier DJ. 2011. The L-type channel antagonist isradipine is neuroprotective in a mouse model of Parkinson's disease. Neurobiology Disease, 43(2): 364-71.
Ismaili L, Refouvelet B, Benchekroun M, Brogi S, Brindisi P et al. 2016. Multitarget compounds bearing tacrine- and donepezil-like structural and functional motifs for the potential treatment of Alzheimer's disease. Program of Neurobiology S0301-0082(15)00162-8.
Kawamoto EM, Vivar C, Camandola S. 2012. Physiology and pathology of calcium signaling in the brain. Frontiers of Pharmacology 3, 61.
Kreye VA, Luth JB. 1975. Proceedings: verapamil-induced phasic contractions of the isolated rat vas deferens. Naunyn Schmiedeberg Archives of Pharmacology 287 (Suppl R): R43.
Li Y, Wu KJ, Yu SJ, Tamargo IA, Wang Y, Greig NH. 2016. Neurotrophic and neuroprotective effects of oxyntomodulin in neuronal cells and a rat model of stroke. Experimental Neurobiology 288: 104-113.
Maroto M, de Diego AM, Albiñana E, Fernandez-Morales JC, Caricati-Neto, A et al. 2011. Multi-target novel neuroprotective compound ITH33/IQM9.21 inhibits calcium entry, calcium signals and exocytosis. Cell Calcium 50, 359–369.
Mohamed T, Shakeri A, Rao PP. 2016. Amyloid cascade in Alzheimer's disease: Recent advances in medicinal chemistry. European journal of Medicinal Chemistry, 113: 258-272.
Moritoki H, Iwamoto T, Kanaya J, Maeshiba Y, Ishida Y, Fukuda H. 1987. Verapamil enhances the non-adrenergic twitch response of rat vas deferens. European Journal Pharmacology, 140: 75–83.
Neher E, Zucker RS. 1993. Multiple calcium-dependent processes related to secretion in bovine chromaffin cells. Neuron, 10: 21–30.
Onozuka H, Nakajima A, Matsuzaki K, Shin RW et al. 2008. Nobiletin, a citrus flavonoid, improves memory impairment and Abeta pathology in a transgenic mouse model of Alzheimer's disease. Journal of Pharmacology Experimental Therapeutics 326(3): 739-44.
Salat D, Noyce AJ, Schrag A, Tolosa E. 2016. Challenges of modifying disease progression in prediagnostic Parkinson's disease. Lancet Neurology, S1474-4422(16)00060-0.
Samochocki M, Höffle A, Fehrenbacher A, Jostock R, Ludwig J et al. 2003. Galantamine is an allosterically potentiating ligand of neuronal nicotinic but not of muscarinic acetylcholine receptors. Journal of Pharmacology Experimental Therapeutics 305, 1024-36.
Sommer N, Loschmann PA, Northoff GH, Weller M, Steinbrecher A, Steinbach JP et. al. 1995. The antidepressant rolipram suppresses cytokine production and prevents autoimmune encephalomyelitis. Nature Medicine, 1: 244–248.
Swart T, Hurley MJ. 2016. Calcium channel antagonists as disease-modifying therapy for Parkinson's disease: Therapeutic rationale and current status. CNS Drugs 30(12): 1127-1135.
Wu CL, Wen SH. 2016. A 10-year follow-up study of the association between calcium channel blocker use and the risk of dementia in elderly hypertensive patients. Medicine (Baltimore) 95(32): e4593.
Xiao L, O'Callaghan JP, O'Donnell JM. 2011. Effects of repeated treatment with phosphodiesterase-4 inhibitors on cAMP signaling, hippocampal cell proliferation, and behavior in the forced-swim test. Journal of Pharmacology Experimental Therapeutics, 338: 641–647.
Zamponi GW. 2016. Targeting voltage-gated calcium channels in neurological and psychiatric diseases. Nature Review Drug Discovery, 15, 19-34.