

Nilesh P. Babre1*, T. Shivaraj Gouda2, Narayanswamy Lachmanan Gowrishankar3
1Department of Pharmacology, VIVA Institute of Pharmacy, Virar (E), Palghar, Mumbai, 401303 India
2Department of Pharmacology, NET Pharmacy College, Raichur - 584103, Karnataka, India
3Department of Pharmacognosy, Prime College of Pharmacy, Erattayal, Kodumbu, Palakkad - 678551, Kerala, India
*Address for Corresponding Author
Nilesh P. Babre
Late Shri Vishnu Waman Thakur charitable Trust`s
VIVA Institute of Pharmacy, At. Shirgaon, Veer Sawarkar Road, Virar (E)
Taluka: Vasai, Dist. Palghar, Mumbai, Maharashtra, India
Abstract
Objective: To study the hepato protective activity of aerial parts of Pentatropis navalis in CCL4 induced rats. Material and methods: Hepatotoxicity was induced by CCL4 (2 ml/kg i. p) in rats. Silymarin was used as a standard drug and Methanol extract of Pentatropis nivalis 200 mg and 400 mg /kg b. wt. were taken for the study. The degree of protection was determine biochemical parameter like SGOT, SGPT, ALP, Cholesterol, Total Bilirubin and Total Protein level in blood serum. Results and Conclusion: The present study revealed that the methanol extract of aerial part of Pentatropis nivalis contained alkaloids, cardiac glycosides, flavonoids, glycosides, phenols, resins, saponins, tannins, steroids and terpenoids. A significant increase in serum SGPT, SGOT and ALP ,Cholesterol, total bilirubin and marked decrease in total protein level was observed in the animals treated with CCL4 (2 ml/kg i. p. ) as compared to normal. Treatment with MEPN (200mg/kg b. wt.) and (400mg/kg b. wt.) for the five days decrease the all the above parameter significantly (P<0.001) Silymarin pretreatment produces significantly (P <0.001) decreases all the parameter when compared with positive control group. The MEPN at the dose of 400 mg/kg b. wt. exhibited significant activity as compared to MEPN at the dose of 200 mg/kg b. wt. Histopathological study also marked that the improvement in hepatocytes is dose dependent manner. Further study need to isolation and characterization of bioactive molecules in the Pentatropis nivalis.
Keywords: Pentatropis nivalis, hepatoprotective, Carbontetrachloride, Silymarin
Introduction
Liver is essential organ of the human body which performs the detoxification of exogenous xenobiotic, drugs, viral infections and chronic alcoholism. The liver is involved with almost all the biochemical pathways to growth, fight against diseases, nutrient supply, energy provision and reproduction (Ward and Daly, 1999). Damage to hepatic parenchymal cells may contribute to physiological roles having a lethal effect (Wolf, 1999). Carbon tetrachloride [CCL4] is the most commonly used hepatotoxin to study on experimental animals (Johnston and Korening, 1998). CCL4 is a chemical toxin, which causes free radical mediated toxicity.(Singh et al., 2008 ) Free radicals induced an oxidative state that can lead to cellular injury with the consequent alteration in metabolic process. Reactive oxygen species (ROS) plays an important role in the pathogenesis of numerous degenerative diseases have been arises like atherosclerosis, liver damage, kidney and lungs damage, diabetes mellitus & aging (Johnston and Korening, 1998). Hepatic damage induced by CCL4 is an increase in Serum Glutamate oxaloacetate Transaminase, Serum Glutamate Pyruvate Transaminase, Alkaline Phosphatase & total bilirubin concentrations. Increases in concentration of SGPT and SGOT are considered to be important markers for hepatic damage (Rabea et al., 2011: Bigoniya et al., 2009).
Herbal drugs plays an important role in the management of various liver disorder and get heals by naturally with speed up. Many medicinal plants and their formulations are used for liver disorder in ethno medicinal practice (Gedara et al., 2003). The Pentatropis nivalis (J. F. Gmel) D. V. Field & J. R. I. Wood belonging to the family Asclepiadaceae. In folklore medicine it is has been documented that Pentatropis nivalis is used for curing piles, inflammation, leucoderma, biliousness (Ayurveda), gonorrhea, emetic, kapha (Kirtikar and Basu, 1987). This plant extract was evaluated for its antioxidant property (Babre et al., 2018) and other species of plants shows anti-inflammatory, analgesis, anti-ulcers were reported. Keeping this view the present study was carried out on methanolic extracts of aerial part of Pentatropis Nivalis (Asclepiadaceae) for its hepatoprotective effect.
Material and methods
Plant collection and authentication
Aerial part of Pentatropis nivalis was collected from Keshavshrushsti, Bhayandar, Mumbai, The collected plant material was authenticated by Dr. Rajendra D. Shinde, Head, Department of Botany & Director, Blatter Herbarium, St. Xavier’s college, Mumbai. (Herbarium Specimen number NI-1922) The collected plant material was washed with water & then samples were air-dried at room temperature with dehumidifier. Dried samples were ground to powder using a mechanical grinder, and stored in a sealed plastic container.
Extraction method
In brief, 2200 g coarse powder of the sample was extracted in soxhlet extractor with methanol and water. Then the extract was evaporated by using rotatory vacuum evaporator to get semisolid mass. The concentrated extracts of aerial part of P. nivalis were stored in air tight close container in dark place (Harbone, 1973).
Phytochemical analysis
Phytochemical study was carried out for Methanolic extract of Aerial Part of P. Nivalis to detect the presence of different class of phytoconstituents like flavonoids, tannins, saponins, glycosides etc. by using standard procedure (Thripati et al., 2010; Ayoola et al ., 2008).
Experimental animals
Healthy Wistar rats (150-180 g) were used from our animal house facility. Animals were housed separately in polycarbonate cage (29 cm × 22 cm ×14 cm) under standard laboratory conditions with Food and water ad libitum, and lights were on from 07:00 to 19:00 h. Experimental protocol was approved by the institutional animal ethics committee, approval no. (IAE/SKIPS/2016/03) and conformed to the Indian National Science Academy guidelines for the use and care of experimental animals in research.
Acute toxicity study and dose findings studies: (OECD Guidelines-423, 2004)
Acute toxicity study for the methanolic extract P. Nivalis was carried out on Mice according to OECD guidelines. Three mice were fasted overnight and maintained with water ad libitum. Each animal received single dose of MEPN (2000 mg/kg, p.o.) was given to each animal. The animals were under observation for 1h and then 6 h and eventually 24 h up to 7 days for any toxicological symptoms and mortality.
Hepatoprotective activity
In the present activity, the dose dependent study of the MEPN was evaluated in albino rats, the rats were randomly assigned in to five groups and each group consists of six individuals’ animals.
Group I: Normal control where the animals were administered 1ml of 2 % acacia solution of P.O. for five days. And olive oil 2ml/kg on 2nd and 3rd day.
Group –II: Positive control (+ ve Control) where the animals were administered 1ml of 2 % acacia solution P.O. for five days.
Group –III: Where the animals were administered MEPN 200 mg/kg b. wt P.O. for five days.
Group-IV: Where the animals were administered MEPN 400 mg/kg b. wt P.O. for five days.
Group -V: Standard control, where the animals were administered with Silymarin 50 mg/ kg. b. wt for five days.
Induction of Hepatotoxicity
Carbon tetrachloride was administrated by Intra peritoneal route by mixing with olive oil (1:1). Upon this 2nd and 3rd day, group II, III, IV and V received carbon tetra chloride at a dose 2ml/kg b. wt. i. p. (Bagban et al., 2012).
Collection of blood Sample
After 30 minute’s drugs treatment. Blood samples were collected on 6th day by retro orbital plexus route for evaluating various biochemical parameters (Yadav et al., 2008) for the assessment of various biochemical parameters, blood was centrifuged at 4000 rpm for 15 min and serum was collected. Serum were used to determine the biochemical parameters like serum glutamic oxalo-acetic transaminase (SGOT), serum glutamate-pyruvate transaminase (SGPT), alkaline Phosphatase (ALP), total bilirubin, cholesterol and total protein using commercially available kits (Span Diagnostics Ltd., Surat, India). Overnight rats were fasted and sacrificed by using ethyl anesthesia at the end of the experiment.
Histopathology
The animal were sacrificed and liver was collected and wash with normal saline water & stored in 10% of formalin solution for histology to evaluate the hepato-architecture. Study was done by dissecting the liver and observed cellular damage to the liver cells and hepatic veins for microscopy. Sections were prepared and stained with hematoxylin (H) and eosin (E) dye for photo microscopic evaluation (100X) including cell necrosis, fat migration, ballooning, degeneration, and central vein infiltration.
Statistical analysis
All the results were Statistical analyzed as Mean ± SEM (n=6) were performed with one way analysis of variance (ANOVA) followed by Bonferroni's multiple comparisons test.
Results
Preliminary qualitative phytochemical analysis
The present study revealed that the methanolic of aerial part of Pentatropis nivalis. The percentage yield of MEPN was found 6.4 g% w/w. The phytochemical analysis of extract contained alkaloids, cardiac glycosides, flavonoids, glycosides, phenols, saponins, tannins, steroids and terpenoids.
Table 1. Phytochemical evaluations of methanol extract of aerial parts of Petatropis navalis
|
S. NO |
Plant Constituents |
MEPN |
|
1 |
Alkaloids |
+ |
|
2 |
Cardiac glycosides |
++ |
|
3 |
Flavonoids |
++ |
|
4 |
Glycoside |
+++ |
|
5 |
Phenols |
++ |
|
6 |
Saponins |
++ |
|
7 |
Steroids |
+++ |
|
8 |
Tannins |
++ |
|
9 |
Terpenoids |
++ |
Acute toxicity studies
The methanol extract of P. Nivalis did not cause any mortality when the dose is administered 2000mg /kg. b. wt. and hence 1/10th and 1/5th of the maximum dose were chosen for the present study.
Hepatoprotective Study
Effect of MEPN on liver enzymes level in CCL4 induced hepatotoxicity rats
A significant increase in serum SGPT, SGOT and ALP level was observed in the animals treated with CCL4 (2 ml/kg i.p) as compared to normal. Pretreatment with MEPN (200mg/kg b.wt.) and (400mg/kg b.wt.) for the five days decrease the all the above parameter significantly (P <0.001) as elicited in table 2 and figure 1. Silymarin pretreatment produces significantly (P <0.001) decreases all the parameter when compared with positive control group.
Table 2. Effect of MEPN on enzyme SGOT, SGPT and ALP level in blood serum of CCL4 induced hepatotoxicity
|
Groups |
Treatment |
Parameters |
||
|
SGOT (U/L) |
SGPT (U/L) |
APL (U/L) |
||
|
Group I |
Normal Control |
113.0±6.923 |
82.33±2.171 |
64.00±2.436 |
|
Group II |
Positive Control(CCL4) |
375.0±4.313 |
238.0±5.132 |
184.0±4.258 |
|
Group III |
MEPN 200 mg/kg |
265.0±5.477** |
183.2±2.587*** |
175.5±3.096** |
|
Group IV |
MEPN 400 mg/kg |
170.8±4.729*** |
122.7±5.840*** |
136.2±1.600*** |
|
Group V |
Standard control (Silymarin ) |
122.5±5.439*** |
100.0±2.595*** |
89.50±3.160*** |
All groups were compared with positive control group. All the values were expressed Mean ± SEM, Values were represented with statically significance ***P <0.001, ** P<0.01, *P<0.05 (where n=6 animals per group) All the values were analyzed by one way ANOVA followed by Bonferroni's multiple comparisons test.
Effect of MEPN on measurement of Cholesterol, total bilirubin and total protein level in
From the observed results a significant increase in serum level Cholesterol, total bilirubin and marked decrease in total protein was observed in the animals treated with CCL4 (2 ml/kg i. p.) as compared to normal. Pretreatment with MEPN (200mg/kg b. wt.) and (400mg/kg b. wt.) for the five days decrease the all the above parameter significantly (P <0.001) as elicited in Table 3 and figure 1. Silymarin pretreatment produces significantly (P <0.001) decreases all the parameter when compared with positive control group. The MEPN at the dose of 400 mg/kg b. wt. exhibited significant activity as compared to MEPN at the dose of 200 mg/kg b.wt.
Table 3. Effect of MEPN on Cholesterol, total bilirubin and total protein level in blood serum of CCL4 induced hepatotoxicity
|
Groups |
Treatment |
Parameters |
||
|
Cholesterol(mg/dl) |
Total Bilirubin(mg/dl) |
Total Protein (g/dl) |
||
|
Group I |
Normal Control |
113.2±0.9098 |
0.2345± 0.01159 |
7.322±0.1823 |
|
Group II |
Positive Control(CCL4) |
212.2±4.622 |
1.720±0.08198 |
5.533±0.1085 |
|
Group III |
MEPN 200 mg/kg |
164.5±2.217*** |
1.597±0.03981ns |
5.660±0.1590ns |
|
Group IV |
MEPN 400 mg/kg |
129.3±3.373*** |
1.423±0.1124** |
6.435±0.1042** |
|
Group V |
Standard control (Silymarin ) |
119.0±1.633*** |
0.2712±0.01522*** |
6.683±0.1470*** |
All groups were compared with positive control group. All the values were expressed Mean ± SEM, values were represented with statically significance ***P <0.001, ** P<0.01, *P<0.05 (where n=6 animals per group) All the values were analyzed by one way ANOVA followed by Bonferroni's multiple comparisons test
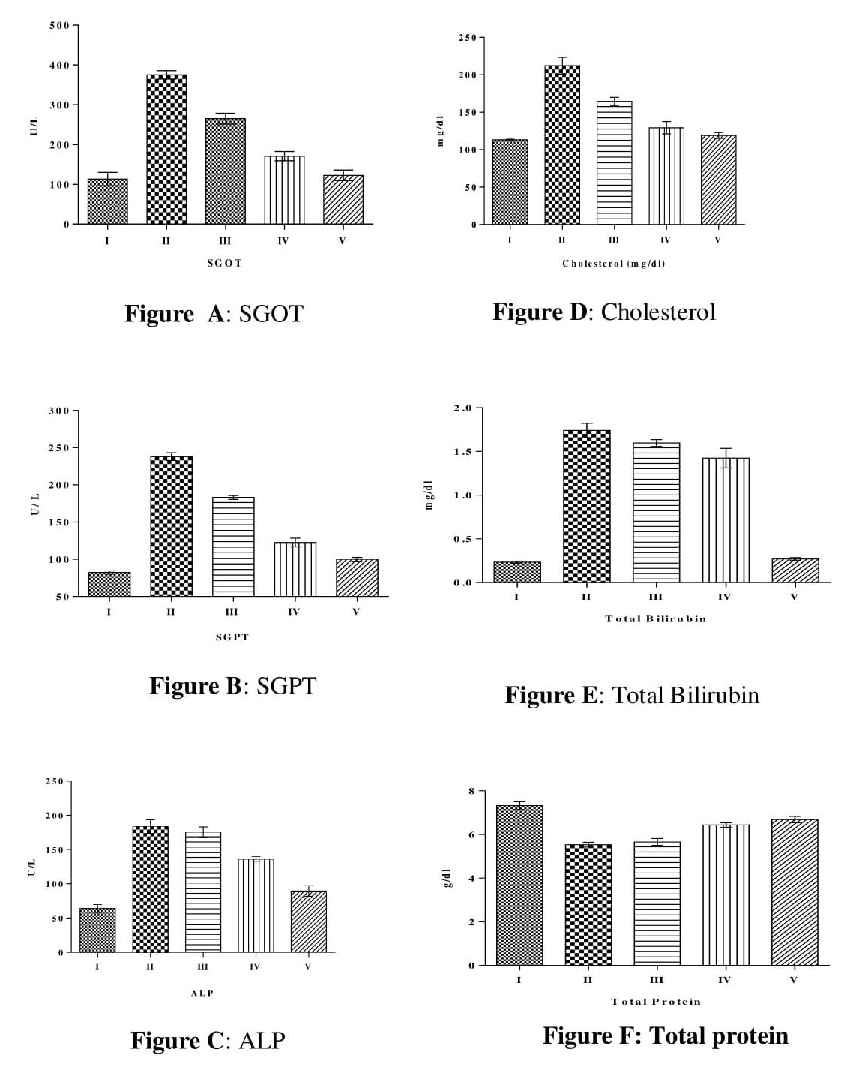
Figure 1. Effect of MEPN on enzyme level of SGOT, SGPT, ALP and measurement of cholesterol, total bilirubin and total protein level in blood serum of
Histopathological observations
Histopathological studies were represented in the figure 2. In the observation group I (A) the liver section of normal rats shows the normal liver lobular structure and no pathological changes were observed in the normal control group. Group II (B) in the positive control i.e. CCL4 treated group shows that there was a degeneration of hepatocytes, necrosis and loss of cellular boundaries. Group III and IV (C, D) after the administration MEPN at the dose of 400 and 200 mg/kg b. wt. these histopathological changes were remarkably reversed and seen with lesser degeneration of hepatocytes. Group V (E) Standard Control (Silymarin) significantly normalized these effects. Silymarin and MEPN 400 mg/kg b. wt. treated group were correlates al the serum biochemical results.
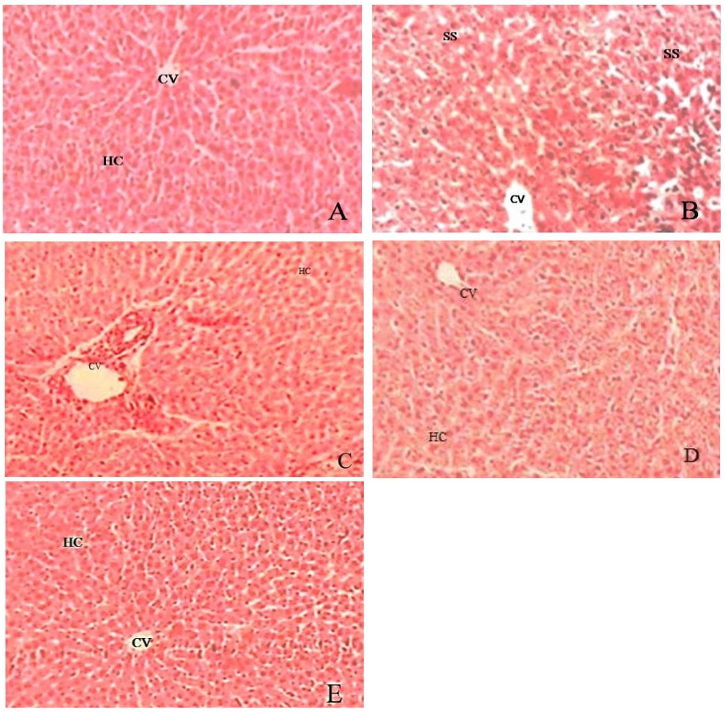
Figure 2. Histopathological architecture in the liver of control and experimental groups (A) Normal Control, (B) Positive control (CCL4), (C) MEPN 200 mg /kg b.wt. (D) MEPN 400 mg /kg b. wt and (E) Standard Control (Silymarin); where CV: Central Vein, HC: Hepatic Cell and SS: Sinusoidal Space
Discussion and Conclusion
Aerial part of Pentatropis Nivalis was collected, authenticated, dried and ground to powder. Further plant material was extracted with methanol by soxhlet method. Phytochemicals screening of was performed which showed the presence of Glycoside, steroids, saponins, flavonoids, phenols and terpenoids. Methanolic extract showed better qualitative tests for presence of secondary metabolites than aqueous extract. The extract was subjected to acute toxicity study according to guide line of OECD 423, no lethality was observed at 2000mg / kg b.wt. Based upon the review of literature finding that P. Nivalis possesses good antioxidant property (Babre et al., 2018). The primary Phytochemical extract TLC (Thin layer Chromatography) shows the presence of flavonoids, terpenoids and phenol class of compound (Babre et al., 2018). The presence of flavonoids and phenol is known for its antioxidant and hepatoprotective capacity (Di Carlo et al., 1999). The hepatoprotective study was conducted on CCL4 induced hepatotoxic in rats. The result was observed in dose dependent manner methanol extract of Pentatropis Nivalis 400 mg/kg b. shows significant results as compared to 200mg /kg b.wt. these finding indicate that the plant possesses good hepatoprotective property when compared to Positive control (CCL4) Histopathological architecture also support to all the biochemical parameter.
References
Bagban IM, Pal SP, Chaudhury A, Das SK, Gohil KJ, Bandari KK. 2012. Hepatoprotective activity of the methanolic extracts of Fagonia indica Burm in carbon tetra chloride induced hepatotoxicity in rats. Asian Pacific Journal of Tropical Biomedicine, 2012 (S1): 245–9.
Bigoniya P, Singh CS, Shukla A. 2009. A comprehensive review of different liver toxicants used in experimental pharmacology. International Journal of Pharmaceutical Sciences and Drug Research, 1(3): 124-135.
Di Carlo G, Mascolo N, Izzo AA, Capasso F. 1999. Flavonoids: old and new aspects of a class of natural therapeutic drugs. Life sciences, 65(4):337-53.
Ayoola GA, Coker HAB, Adesegun SA, Adepoju-Bello AA, Obaweya K, Ezennia EC, et al. 2008. hytochemical screening and antioxidant activities of some selected medicinal plants used for malaria therapy in Southwestern Nigeria. Tropical Journal of Pharmaceutical Sciences, 7 (3): 1019-1024.
Gedara SR, Abdel-Halim OB, El-Sharkawy SH, Salama OM, Shier TW, Halim AF. 2003. New erythroxane-type diterpenoids from Fagon boveana (Hadidi) & Graf. Zeitschrift fur Naturforschung, 58: 23-32.
Harbone JB. Phytochemical Methods. 1973. 1st Ed. Chapman and Hall, London. 1973; 60-66.
Johnston DE, Keening C. 1998 Mechanisms of early carbon tetrachloride toxicity in cultured rat hepatocytes. Pharmacology and Toxicology, 83, 231-239.
Kirtikar KR, Basu BD. 1987. Indian medicinal plants, 2ndEds, Dehradun, India, International book distributors and book sellers, pp 1614-15.
Kumar SVS, Mishra SS. 2006. Hepatoprotective effect of extracts from Pergularia daemia Forsk. Journal of Ethno Pharmacology, 107(2):164–8.
Babre NP, Gouda TS, Gowrishankar NL. 2018. Phytochemical Composition and In Vitro Antioxidant Activity of Methanolic And Aqueous Extracts Of Aerial Part Of Pentatropis Nivalis (Asclepiadaceous). International Journal of Phytomedicine, 10(1): 68-72.
Parveen R, Baboota S, Ali J, Ahuja A, Vasudev SS, Ahmad S. 2011. Effects of silymarin nanoemulsion against carbon tetrachloride-induced hepatic damage. Archives of Pharmacal Research, 34 (5): 767-774.
Singh N, Kamath V, Narasimhamurthy K, Rajini PS. 2008. Protective effects of potato peels extract against carbon tetrachloride induced liver injury in rats. Environmental Toxicology and Pharmacology 26, 241–246.
Tripathi RC, Pandey MB, Singh S, Pandey VB. 2010. Antifungal flavonoids of Melochia corchorifolia. Journal of the Indian Chemical Society, 87(4), 511-512.
Ward FM, Daly MJ. Hepatic Disease. In: Clinical Pharmacy and Therapeutics, Walker R. and C. Edwards Eds. Churchill Livingstone, New York; 1999 pp. 195-212.
Wolf PL. Biochemical diagnosis of liver disease. 1999. Indian Journal of Clinical Biochemistry. 14(1):59-90.
Yadav NP, Pal A, Shanker K, Dyaneshwar UB, Guptan AK, Darkar MP, Suman PSK. 2008. Synergistic effects of silymarin and standardized extract of Phyllanthus amarus against CCl4 induced hepato toxicity in ruttus norvegicus. Phytomedicine, 15:1053–61.