

Vikrant K. Nikam1*, Ankita Ugale1, Jyoti Khapare2
1Amrutvahini College of Pharmacy, Sangamner 422 608, (MS) India
2Dr. Naikwadi college of Pharmacy, Jamgaon Sinnar 422 113, (MS) India
*Address for Corresponding Author
Vikrant K. Nikam
Amrutvahini College of Pharmacy, Sangamner (M.S.) 422608, India
Abstract
In 20th century 1970s and 80s when the first thorough fundamental studies in the united states and japan researchers used the term ultrafine particles. However during the 1990s before the national nanotechnology initiative was launched in the United States the term nanoformulation had become more common. Drug delivery system is defined by national institute of health in USA as, “Formulation of a device that enables the introduction of therapeutic substances in to the body and improves efficiency and safety by controlling the rate, time and place of release of drug in the body. Conventional Drug delivery system has many drawbacks including poor bioavailability, side effects, low drug loading capacity, plasma fluctuation of the drug levels, low therapeutic effectiveness, and lack of target delivery. For example, in cancer treatment the conventional drug delivery to the tumor cells can affect normal tissues causing nephrotoxicity, neurotoxicity, cardio toxicity. These drawbacks have motivated on scientists to investigate more about new Drug delivery system. The chemical and physical properties of nanoparticles make then efficient Drug delivery system that have the potential to improve the bioavailability, drug carrying capacity, stability for the drugs within the body. Novel drug delivery system is a combination of advance technique and new dosage form which are better than conventional dosage form. Basic modes of novel drug delivery system are Targeted drug delivery system, controlled drug delivery system which is used in pharmaceutical science. The present reviews gives information regarding various novel techniques used for improving safety and efficacy of phytomedicines and application of novel formulation.
Keyword: Novel drug delivery nanoformulation, stability, impurity
Introduction
Nano-formulations of medicinal drugs have attracted the interest of many researchers for drug delivery applications. These nanoformulations enhance the properties of conventional drugs and are specific to the targeted delivery site (Jeevanandam et al., 2016).
In 19th century Michael faraday provided the first description in scientific terms, of the optical properties of nanometer-scale metals in his classic 1857 paper in subsequent paper, the author point out that it is well known that when thin leaves of gold or silver are mounted upon glass and heated to a temperature that is well below a read heat (~500°C), a remarkable change of properties takes place, whereby the continuity of the metallic film is destroyed.
In 20th century 1970s and 80s when the first thorough fundamental studies in the united states and japan researchers used the term ultrafine particles. However during the 1990s before the national nanotechnology initiative was launched in the United States, the term nanoformulation had become more common (Yadav et al., 2012).
Nanotechnology is a rapidly expanding field due to the multidisciplinary support from researchers in the academic, industry, and federal sectors. Nanotechnology plays an important role in therapies of the future as nanomedicines by enabling this situation to happen, thus lowering doses required for efficacy as well as increasing the therapeutic indices and safety profiles of new therapeutics (Genç et al., 2011).
Drug delivery system is defined by national institute of health in USA as, “Formulation of a device that enables the introduction of therapeutic substances in to the body and improves efficiency and safety by controlling the rate, time and place of release of drug in the body. More than 40% of drugs are poorly soluble in water, so they show problems in formulating them in conventional dosage forms. Also, for class II drugs which are poorly soluble in aqueous and organic media, the problem is more complex. Conventional Drug delivery system has many drawbacks including poor bioavailability, side effects, low drug loading capacity, plasma fluctuation of the drug levels, low therapeutic effectiveness, and lack of target delivery. Traditional Drug delivery system circulate drugs to the cells in body non-selectively, which can lead to serious consequences such as side effects, multiple drug resistance and reduced drug concentration at target location (Yadav et al., 2012).
For example, in cancer treatment the conventional drug delivery to the tumor cells can affect normal tissues causing nephrotoxicity, neurotoxicity, cardiotoxicity. These drawbacks have motivated on scientists to investigate more about new Drug delivery system.
How Nano-technology can overcome these drawbacks can be understood by discussing the mechanism of drug delivery using Nano-particles? The process of drug delivery can be mainly divided as; the administration of the drug or therapeutic product; release of the active part of the drug and transport active ingredients across the biological membrane to the target site to perform action. The chemical and physical properties of nano particles make then efficient Drug delivery system that have the potential to improve the bioavailability, drug carrying capacity, stability for the drugs within the body, controlled release, and targeted delivery (Witharana et al., 2017).
Bioavailability refers to the extent and rate at which the active moiety enters systemic circulation, thereby accessing the site of action. Nano-technology increases bioavailability of drugs as a result of their special uptake mechanisms such as absorptive endocytosis and the ability to avoid degradation in the gastrointestinal track. The drug incorporated in to the nano particle is easily diffused through biological membranes. Drug-polymer attachment changes the drug solubility, hydrophobicity, and permeability. The drug loading capacity can be increased by minimizing solubility, increasing ionic interactions between drug and matrix and by maximizing the absorption of drug load. The nano particle are also able to remain in the blood for long period. The drugs attached nano particle can avoid being attacked by the immune system by having a particle surface decorated with biodegradable, hydrophilic copolymers. Poly- glycolic acid, poly-lactic acid and their co-polymers are widely used for decorating the surface (Witharana et al., 2017).
The self-controlling system of drug release helps to reduce the plasma fluctuation and minimize the side effects. Controlled drug releasing in particular sites can be controlled by different ways,
1) Polymers are biodegradable and it is degraded in controllable manner to release drugs to the site.
2) Pores within the polymer can be altered so that drug diffusion occurs more readily or slowly.
3) The distance of fusion and surface area of the nano particle can be altered by changing size. Smaller size means larger surface area and drug release and dissolving becomes faster. The drugs are released by matrix by diffusion, swelling, erosion or degradation. The drug release is controlled by osmotic pressure, mechanical pumping and through electro kinetic transport. Nano-technology based drug delivery system provides drugs to target sites by their ligands attraction process (Rangasamy et al., 2010).
The Nano particle surface can be decorated by ligands and these ligands can attach to the specific receptors in the surface of targeted cell by bio-recognition. The nano particles enter the target cells by receptor mediated endocytosis. Inside the cell nano particles are developed in to endosomes. Then endosomes merge with each other to form large endosomes or lysosomes. Finally, therapeutic drugs can be released in response to enzymes or acidic pH with controllable manner by degradation of polymeric nano particles shell. Ultimately nano particles in drug delivery system enhance the ability to use highly toxic, poorly soluble, unstable drugs and maximizing patient comfort (Witharana et al., 2017).
The method by which a drug is delivered can have a significant effect on its efficacy. Some drugs have an optimum concentration range within which maximum benefit is derived, and concentrations above or below this range can be toxic or produce no therapeutic benefit at all. On the other hand, the very slow progress in the efficacy of the treatment of severe diseases, has suggested a growing need for a multidisciplinary approach to the delivery of therapeutics to targets in tissues. From this, new ideas on controlling the pharmacokinetics, pharmacodynamics, non-specific toxicity, immunogenicity, bio recognition, and efficacy of drugs were generated. These new strategies, often called drug delivery systems (DDS), are based on interdisciplinary approaches that combine polymer science, pharmaceutics, bio-conjugate chemistry, and molecular biology. Dramatic changes have in introduced, with new technology and new devices now on market. In some cases traditional capsules and ointments have been replaced by osmotic pumps, wearable ambulatory pumps, electrically assisted drug delivery and host of other delivery methods based on various polymer technologies. In some cases the new drugs require new delivery systems because the traditional systems are inefficient and ineffective. Some therapies may become very site specific and require very high concentrations of drugs in selected sites of body, as more Controlled drug delivery systems will be available very near future. New drug delivery system development is largely based on promoting the therapeutic effects of a drug and minimizing its toxic effects by increasing the amount and persistence of a drug in the vicinity of target cell and reducing the drug exposure of non-target cells (Yadav et al., 2012).
Novel drug delivery systems can include those based on physical mechanisms and those based on biochemical mechanisms. Physical mechanisms also referred as controlled drug delivery systems include osmosis, diffusion, erosion, dissolution and electro transport. Biochemical mechanisms include monoclonal antibodies, gene therapy, and vector systems, polymer drug addicts and liposome.
Therapeutic benefits of some new drug delivery systems include optimization of duration of action of drug, decreasing dosage frequency, controlling the site of release and maintaining constant drug levels. Among drug carriers one can name soluble polymers, micro particles made of insoluble or biodegradable natural and synthetic polymers, microcapsules, cells, cell ghosts, lipoproteins, liposome, and micelles.
The carriers can be made slowly degradable, stimuli-reactive and even targeted. Targeting is the ability to direct the drug-loaded system to the site of interest (Rangasamy et al., 2010).
Novel carrier for controlled and targeted drug delivery
As the knowledge of the molecular biology and pathophysiology of diseases has expanded, more therapeutically précised and purpose specific drug are being developed. These newly developed drug have high potency (low therapeutic window) and required their localization of the particular site of their action. Most drugs are administrated by conventional immediate- release dosage forms. They distribute freely throughout the body and accumulate the non – specific organs in an undesirable manner and thus produce adverse side effects. To reduce these slides and increased their therapeutic benefits, they should be delivered to their respective site of action, and hence suitable carrier systems becomes mandatory requirement. Various novel carriers have been developed for the purpose. Among these colloidal carriers such as liposome’s, nano- particles & supra molecular system, i.e. micelles have gained more attention in the field of controlled and targeted drug delivery. Recently new carriers such as inorganic particles, liquids crystal, aquasomes, carbon nano tubes, dendrimers etc. Are also investigated for the specialized purpose. In the following section, these carriers for the same purpose are brief (Khan et al., 2017).
Nanosome: Non – ionic surfactants vesicles (nanosomes) are now widely studied as an alter -native to liposomes. Non -ionic surfactants vesicles results from the self- assembly of hydrated surfactants monomers. Non – ionic surfactants of wide variety of structural types have been found to be useful alternatives to phospholipids. Through the terminology suggests that distinctions exist between nanosomes and liposomes of which the former is having chemical differences in the monomers units, nanosomes posses physical properties, which are similar to liposomes, which are formed from phospholipids. As the name indicated, generally non- ionic surfactants vesicles are prepared by the incorporation of components containing non- ionic surfactants. However, they may also prepared with various ionic amphiphiles such as dicetyl phosphate, stearyl amine, etc. in order to achieve a stable vasicular suspension. It is important to identify and know the basic structural units of NSVs. while an amphiphilic head groups. The vesicles forming non-ionic compounds are mainly alkyl ether lipids. These can be broadly divided into two classes based on nature of their hydrophilic head groups, i.e. Alkyl ethers in which the hydrophilic head group consists of repeat glycerol subunits, related isomers or larger sugar molecules, and those in which the hydrophilic head group consists of repeat ethylene oxide subunits. In addition, alkyl esters, amides and fatty acids, and amino acids compounds also from vesicles (Khan et al., 2017).
Liposome: Liposome’s were first produced in England in 1961 by Alec D. Bangham. Liposome’s are colloidal, concentric layered vesicles in which an aqueous volume is entirely enclosed by membranous lipid bilayers mainly composed of natural or synthetic phospholipids. The polar character of the liposomal core enables polar drug molecules to be encapsulated. The ability of liposome to extra vasate and penetrate into diseased tissues is directly correlated with their size. Large liposome are rapidly removed from circulation into tissues of macrophage origin and do not achieve significant level in other tissues of the body. The development of techniques for making liposome populations of homogeneous small size (≤100 nm). Amphiphilic and lipophilic molecules are solubilised within the phospholipids bilayers according to their affinity towards the phospholipids. Participation of nonionic surfactants instead of phospholipids in the bilayers formation results in niosomes. Channel proteins can be incorporated without loss of their activity within the hydrophobic domain of vesicle membranes, acting as a size-selective filter, only allowing passive diffusion of small solutes such as ions, nutrients and antibiotics (Bhagwat et al., 2013; Sharma et al., 1997).
Thus, drugs that are encapsulated in a nanocage-functionalized with channel proteins are effectively protected from premature degradation by proteolytic enzymes. The drug molecule, however, is able to diffuse through the channel, driven by the concentration difference between the interior and the exterior of the nanocage. Dendrimers are nanometer-sized, highly branched and monodisperse macromolecules with symmetrical architecture. They consist of a central core, branching units and terminal functional groups. The core together with the internal units, determine the environment of the nano cavities and consequently their solubilizing properties, whereas the external groups the solubility and chemical behavior of these polymers. Targeting effectiveness is affected by attaching targeting ligands at the external surface of dendrimers, while their stability and protection from the Mononuclear Phagocyte System is being achieved by functionalization of the dendrimers with polyethylene glycol chains (PEG). Liquid Crystals combine the properties of both liquid and solid states. They can be made to form different geometries, with alternative polar and non-polar layers (i.e., a lamellar phase) where aqueous drug solutions can be included (Allen et al., 1996; Tiwari et al., 2012; Srinivas et al., 2015).
Niosomes: In niosomes, the vesicles forming amphiphile is a non-ionic surfactant such as Span – 60 which is usually stabilized by addition of cholesterol and small amount of anionic surfactant such as dicetyl phosphate. Niosomes and liposomes are equiactive in drug delivery potential and both increase drug efficacy as compared with that of free drug. Niosomes are preferred over liposomes because the former exhibit high chemical stability and economy. Surfactant forming niosomes are biodegradable, non-immunogenic and biocompatible. niosomes have properties as potential drug carriers similar to liposomes. Niosomes are different from liposomes in that they offer certain advantages over liposomes (Bhagwat et al., 2013; Bandawane et al., 2019; Madhav et al., 2011).
Figure 1. List of drug carrier in NDDS
Nanoparticle: Nanoparticles are submicron-sized polymeric colloidal particles with a therapeutic agent of interest encapsulated within their polymeric matrix or ad-sorbed or conjugated onto the surface. We have been investigating nanoparticles formulated using a FDA approved biodegradable and biocompatible polymers. Therapeutic agent encapsulated into the poly-mer for localized and sustained drug and macro-molecular delivery.
Nanoparticles (including nanospheres and nanocapsules of size 10-200nm) are in the solid state and are either amorphous or crystalline. They are able to adsorb and encapsulate a drug, thus protecting it against chemical and enzymatic degradation. In recent years, biodegradable polymeric nanoparticles have attracted considerable attention as potential drug delivery devices in view of their applications in the controlled release of drugs, in targeting particular organ or tissue, as carriers of DNA in gene therapy, and in their abilities to deliver proteins peptides and genes through peroral route (Bhagwat et al., 2013; Panyam et al., 2003).
Example: Nanoparticle delivery to the lungs has many challenges including formulation instability due to particle–particle interactions and poor delivery efficiency due to exhalation of low-inertia nanoparticles. Thus, novel methods formulating nanoparticles into the form of micron-scale dry powders have been developed (Sung et al., 2007).
Microspheres: Microspheres are characteristically free flowing powders consisting of proteins or synthetic polymers which are biodegradable in nature and ideally having a small spherical particles, with diameters in the micrometer range (typically 1 μm to 1000 μm (1 mm)).A well designed controlled drug delivery system can overcome some of the problems of conventional therapy and enhance the therapeutic efficacy of a given drug. Microspheres provide constant prolonged therapeutic effect. There are various approaches in delivering a therapeutic substance to the target site in a sustained controlled release fashion. Materials used for preparing Microspheres are polymers (Bhagwat et al., 2013). They are classified into two types:
Microspheress received much attention not only for prolonged release, but also for targeting of anticancer drugs to the tumour (Thakkar et al., 2004).
1. Synthetic Polymer: Poly alkyl cyano acrylates is a potential drug carrier for parenteral as well as other ophthalmic, oral preparations. Poly lactic acid is a suitable carrier for sustained release of narcotic antagonist, anti cancer agents such as cisplatin, cyclo phosphamide, and doxorubicin. Sustained release preparations for anti malarial drug as well as for many other drug has been formulated by using of co-polymer of poly lactic acid and poly glycolic acid as Poly anhydride microspheres. Poly adipic anhydride is used to encapsulate timolol maleate for ocular delivery. Poly acrolein microspheres are functional type of microspheres (Bhagwat et al., 2013; Khar et al., 2002).
2. Natural polymers:-Albumin is a widely distributed natural protein. It is considered as a potential carrier of drug or proteins (for either their site specific localization or their local application into anatomical discrete sites). It is being widely used for the targeted drug for the targeted drug delivery to the tumors cells (Nikam et al., 2012; Meghna et al., 2017; Betala et al., 2017).
Microparticles: The “microcapsules” are defined as a spherical particle with size varying from 50 nm to 2nm, containing a core substance. Microspheres are, in real sense, spherical empty particles. However, the term microcapsules & microspheres are often used interchangeably. In addition some related terms are used as well for example, “microbeads” & “beads” are used alternatively. Sphere and spherical particles are also used for particles of large size & rigid morphology. The dried microspheres from free flowing powders .they consist of proteins or synthetic polymers, which bio degradable & ideally have a size range less than 200µm. The solid bio degradable microspheres bearing a drug dispensed or dissolved throughout particles matrix have potential in controlled- release of drugs (Khan et al., 2017).
These carriers received much attention not only prolonged release formulations but also for the carrier potential in drug targeting particularly anti- cancer drugs the tumour.
Pre-requisites for ideal micro particulate carriers are follows.
•Longer duration of action
•control of drug release
•Increase of therapeutic efficiency
•Protection of drug
•Biocompatibility
•Relative stability
•water -solubility or Dispensability
•Bioresorbability
•Targetability
•Polyvalency (Khan et al., 2017)
Microemulsion: The microemulsion concept was introduced as early as the 1940s by Hoar and Schulman who generated a clear single-phase solution by titrating a milky emulsion with hexanol. Microemulsions are thus defined as a system of water oil and amphiphile which is a single optically isotropic and thermodynamically stable liquid solution. Microemulsions are clear, stable, isotropic mixtures of oil, water and surfactant, frequently in combination with a co surfactant. These systems are currently of interest to the pharmaceutical scientist because of their considerable potential to act as drug delivery vehicles by incorporating a wide range of drug molecules. In order to appreciate the potential of microemulsions as delivery vehicles. Microemulsions have been studied for delivery of anti hypertensive drug such as lacidipine a poorly water soluble and low bioavailable drug after transdermal administration of microemulsion gel compared to oral suspension. Microemulsion based transdermal therapeutic system of lacidipine was developed and optimized (Khan et al., 2017; Lawrencea et al., 2000).
Nanosuspension: A pharmaceutical nanosuspension is defined as “very finely dispersed solid drug particles in an aqueous vehicle, stabilized by surfactants, for either oral and topical use or parentral and pulmonary administration, with reduced particle size, leading to an increased dissolution rate and therefore improved bioavailability”. The diameter of the suspended particle is less than 1μm in size (i.e. 0.1nm-1000 nm). The particle size distribution of the solid particles in nanosuspensions is usually less than one micron with an average particle size ranging between 200 and 600 nm. An increase in the dissolution rate of micronized particles (particle size < 10μm) is related to an increase in the surface area and consequently the dissolution velocity. Nano size particles can increase dissolution velocity and saturation solubility because of the vapor pressure effect. Class II drugs which are poorly soluble in aqueous and organic media, the problem is more complex. Preparing nanosuspension is preferred for such compounds that are insoluble in water (but are soluble in oil) with high log P value. Various approaches to resolve problems of low solubility and low bioavailability micronization, co-solvency, oily solution, salt formation- some other techniques are liposomes, emulsions, micro emulsion, solid dispersion, ß- cyclodextrin inclusion complex etc. But, many of these techniques are not universally applicable to all drugs. In these cases nanosuspensions are preferred. In case of drugs that are insoluble in both water and in inorganic media instead of using lipidic systems, nanosuspensions are used as a formulation approach (Yadav et al., 2012).
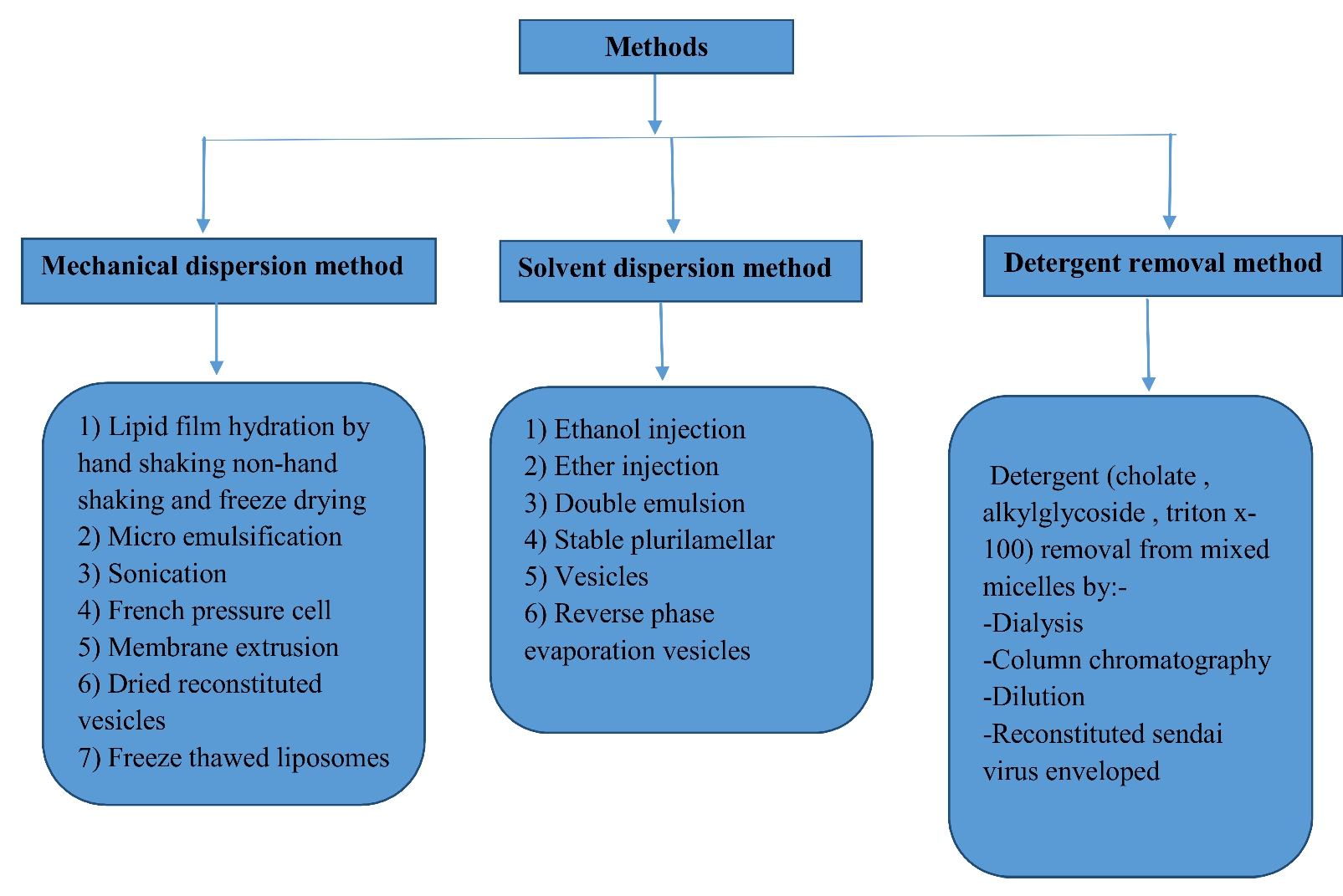
Figure 2. Method of Preparation of NDDS
A nanosuspension not only solves the problem of poor solubility and bioavailability but also alters the pharmacokinetics of drug and that improves drug safety and efficacy. Nanosuspensions differ from nanoparticles, which arepolymeric colloidal carriers of drugs (Nanospheres and nanocapsules), and from solid lipid nanoparticles (Prabhakar, 2011).
Advantages
1. Protection from physical and chemical degradation
2. Sustained delivery
3. Improved tissue macrophages distribution
4. Enhancement of stability
5. Enhancement of pharmacological activity
6. Protection from toxicity
7. Increased bioavailability
8. Enhancement of solubility
9. Better treatment of many chronic illnesses. eg. Cancer, Asthma, Arthritis
10. Increased Bio-availability (Kumar et al., 2011)
Impurities: Impurity is defined as any substance coexisting with the original drug, such as starting material or intermediates or that is formed, due to any side reactions. Impurity can be of three types: Impurities closely related to the product and coming from the chemical or from the biosynthetic route itself, Impurities formed due to spontaneous decomposition of the drug during the storage or on exposure to extreme conditions, or the precursors which may be present in the final product as impurities. Impurities present in excess of 0.1% should be identified and quantified by selective methods. The suggested structures of the impurities can be synthesized and will provide the final evidence for their structures, previously determined by spectroscopic methods. Therefore it is essential to know the structure of these impurities in the bulk drug in order to alter the reaction condition and to reduce the quantity of impurity to an acceptable level. Isolation, identification and quantification of impurities help us in various ways, to obtain a pure substance with less toxicity and, safety in drug therapy. This review covers the study of impurities present in new drug substances (Prathap et al., 2013).
Types of Impurity: Types of Impurity Organic medicinal substances are contaminated in exactly the same manner as inorganic substance during their manufacturing processes. Since the organic substances belong to a very wide range of chemical groups and at the same time the contaminating impurities being of varied nature the task of detecting the impurities becomes a difficult job. Therefore, the contaminating impurities for organic medicinal compounds can be classified into:
Organic Impurities: The actual and potential impurities most likely to arise during the synthesis, purification, and storage of the drug substance should be summarized, based on sound scientific appraisal of the chemical reactions involved in the synthesis, impurities associated with raw materials that could contribute to the impurity profile of the drug substance. The laboratory studies conducted to detect impurities in the drug substance, which include test results of materials manufactured during the development process and batches from the commercial processes. The impurity profile of the drug lots, intended for marketing should be compared with those used in development. The spectroscopic studies (NMR, IR, MS etc. ) conducted to characterize the structure of actual impurities present in the drug substance above an apparent level of 0.1% (e.g., calculated using the response factor of the drug substance) should be described. All recurring impurities above an apparent level of 0.1% in batches manufactured by the proposed commercial process should be identified of these studies (Parimoo et al., 1998).
Inorganic Impurities: Inorganic impurities are normally detected and quantified using Pharmacopeias or other appropriate standards. Carryover of catalysts to the drug substance should be evaluated during development.
Sources of Pharmaceutical Impurity: Impurities associated in with APIs Organic impurities, Inorganic impurities, Solvent residues Impurities related to formulation Impurity forms during formulation, Formation of impurities on aging. Medicines are the formulated forms of active pharmaceutical ingredients. There are 2 types of impurities in medicines: (1) Impurities associated in with active pharmaceutical ingredients and (2) Impurities that form are created during formulation and or with aging or that are related to the formulated forms (Prathap et al., 2013).
Process parameter effects on novel drug delivery system
1) Polymer solubility: In the control release of a drug species for either polymer membrane permeation controlled or polymer matrix diffusion controlled drug delivery devices or other rate programmed drug delivery devices, the drug particles are visualized as not being releasable from the device until the drug molecule on the outer most surface layer of a drug particle
This suggests that the of drug species in rate controlled polymer membrane or matrix plays rate controlling role in its release from a polymeric device (Chein, 2010).
2) Solution solubility: Various studies stated that the release of drugs from controlled device is truly influenced by its solution solubility.
As in vivo sink condition is effectively maintained by active hemo-perfusion, in vitro drug release studies should be conducted under perfect sink condition so the release of drug is controlled solely by delivery system and is not affected by solution solubility factor.
Aqueous solubility varies similar to that of polymer solubility which is very much dependent upon the difference in their chemical structure, type and physicochemical nature of the functional groups and stereo chemical configuration (Chein, 2010).
3) Partition coefficient
The partition coefficient K of a drug for its interfacial partitioning from the surface of drug delivery device toward an elution medium is defined in equation.
K = Cs / Cp
Where, Cs = solubility in elution solution
Cp = solubility in polymer composition
The effect of K on controlled release of drug from a matrix type delivery device was reported to be biphasic both the mechanism and the rate profile of drug release were dependent upon the variation in K.
4) Polymer diffusivity: The diffusion of small molecules in a polymer structure is an energy activated process in which the diffusant molecule move to a successive series of equilibrium position when a sufficient amount of energy, called the energy of activation for diffusion Ed,
5) Solution diffusivity: The diffusion of solute molecule in a solution medium may be considered to result from the random motion of molecules.
Under concentration gradient molecule diffuse spontaneously from a region of higher concentration to region of lower concentration until equilibrium is reached.
6) Thickness of polymer diffusion path:
7) Thickness of hydrodynamic diffusion layer:
The rate limiting role of hydrodynamic diffusion layer HD in determining drug release profile can be visualized by considering the device immersed in stationary position in a solution, a stagnant layer is established on the immediate surface of the device.
8) Drug loading:
In the preparation of drug delivery devices various loading dose of drug in incorporated into the device as required for different length of treatment.
Variation in drug loading dose in matrix type device affects the magnitude of drug release.
Rate of drug release from membrane permeation controlled reservoir type polymer device is independent of drug loading dose.
9) Surface area: Rate of drug release is dependent upon the surface area of drug delivery device both in vivo and in vivo rates of drug release are observed to be upon the surface area of drug delivery device (Chein, 2010).
Stability of Nanoformulation
Drug Stability
Drug stability means the ability of the pharmaceutical dosage form to maintain the physical, chemical, therapeutic and microbial properties during the time of storage and usage by the patient.
Stability studies ensuring the maintenance of product quality, safety and efficacy throughout the shelf life are considered as pre-requisite for the acceptance and approval of any pharmaceutical product. These studies are required to be conducted in a planned way following the guidelines issued by ICH, WHO and or other agencies. Importance of various methods followed for stability testing of pharmaceutical products, guidelines issued for stability testing (Kaur et al., 2013).
Stability is defined as the capacity of drug substance or drug product to remain within the established specification to maintain its identity, strength, quality and purity throughout there test or expiration dating period. Stability testing of pharmaceutical products is a complex set of procedures involving considerable time, cost and scientific expertise in order to build in quality, efficacy and safety in drug formulations. Stability studies are the one of the most important step during the drug development process because it required to assure the identity, potency and purity of ingredients, as well as those of formulated product (Singh et al., 2000).
WHO states that, the stability of finished pharmaceutical products depends on environment factors such as ambient temperature, humidity and light as well as on the product related factors e.g chemical and physical properties of active substance and of pharmaceutical excipient, the dosage form and its composition, the manufacturing process, the nature of the container closure system and properties of packaging material (Tangriet al., 2012).
Type of stability study
Physical stability studies: The physical changes can have deleterious effects too. The physical evaluation of the solution is of particular importance for intrathecal, ocular and intra-arterial routes. A tablet may become soft and ugly or it intra-arterial routes. A tablet may become soft and ugly or it may become very hard and show very slow dissolution time as a result of which bioavailability may not be good, so physical stability studies are also essential. A more refined physical evaluation, using turbidimetry, light odstruction, dynamic light scattering or microscopic analysis, is particularly important for therapeutic proteins to evaluate their kinetic profiles of aggregation.
Chemical stability studies: Moisture take part as a reactant in many chemical reactions and play the role of solvent vector in many reactions. It has better thermal conductivity than solids and allow better heat transfer hence molecules have more kinetic energy and more decomposition is observed. In all these, hydrolysis or oxidation or fermentation; common cause is moisture. All reactions speeded upon presence of moisture. The methods used for evaluation of chemical instability are HPLC, HPTLC or capillary electrophoresis. These methods are used widely.
Microbiological stability studies: Microorganisms not only contaminate formulations containing moisture but also solid dosage forms containing natural polymer because many natural polymers are source of microorganism (Bardin et al., 2011).
Factors affecting drug stability
So if the pH of a drug solution has to be adjusted to improve solubility and the resultant pH leads to instability then a way out of this tricky problem is to introduce a water miscible solvent into the product.
It will increase stability by:
3. Moisture:-Water catalyses chemical reactions as oxidation, hydrolysis and reduction reaction water promotes microbial growth.
Conclusion
Novel drug delivery system is a combination of advance technique and new dosage form which are better than conventional dosage form. Novel drug delivery system overcome non compliances, by reduced the repeated administration of drug, but also helps to increase the therapeutic value by reducing toxicity and increase the bioavailability. Basic modes of novel drug delivery system are Targeted drug delivery system, controlled drug delivery system which is used in pharmaceutical science. In the future it may be possible to develop nano technology treat different disease at the same time by producing multifunctional nano-particles.
Acknowledgement
Authors are thankful to Amrutvahini College of Pharmacy, Sangamner for providing necessary facility to carry out the work.
References
Allen TH, Moase EH. 1996. Therapeutic Opportunities For targeted liposomal drug delivery.Elsevier science Advanced Drug Delivery Reviews, 21:117-133.
Bhagwat RR, Vaidhya IS.2013.Novel Drug Delivery System: An Overview. International Journal of Pharmaceutical Sciences and Research, 4(3): 970-982.
Betala S, 2017. Formulation and evaluation of sustained release microspheres of Metoprolol. International Research Journal of Pharmacy, 8(11):103-108.
Bandawane A, Saudagar R. 2019. A Review on Novel Drug Delivery System: A Recent Trend. Journal of Drug Delivery and Therapeutics. Journal of Drug Delivery & Therapeutics, 9(3):517-521.
Bardin C, Astier A, Vulto A, Sewell G, Vigneron J, Trittler R, Paul M, Tronjniak M and Pinguet F. 2011. Guidline for the practical stability studies of anticancer drugs:a european consensus conference. Annales Pharmaceutiques Francaises, 69:221-231.
Chein YW. 2010. Novel drug delivery system. 2nd ed., rev. New York : M. Dekker, 1992.
Genç L, Dikmen G, Guney G. 2011. Formulation of Nano Drug Delivery Systems. Journal of Materials Science and Engineering, 132-137.
Hunter CA. 1988. Vesicular System (Niosomes and Liposomes) for Delivery of Sodium Stibogluconate in experimental murine visceral leishmaniasis. Journal of Pharmacy and Pharmacology, 1988, 161-164.
Jeevanandam J, Chan Y, Danquah M. 2016. A Review on Nano-formulations of drugs: Recent developments, impact and challenges. Biochimie, 128-129:99-112.
Khar RK, Vyas SP. 2002. Targeted and Controlled Drug Delivery- Novel Carrier Systems., 1st edition, CBS Publications and Distributors, New Delhi,417-418.
Khan MG. 2017. Topic – The Novel Drug Delivery System, World journal of pharmacy and pharmaceutical sciences 6(7): 477-487.
Kumar V, Sharma A, Sharma A, Joshi G, Dhillon V. 2011. Recent advances in novel drug delivery system for delivery of anti- hypertensive drugs. International Journal of Drug Development & Research, 3(1):0975-9344.
Kaur M, Kaur G, Kaur H. 2013. Overview on stability Studies. International Journal of Pharmaceutical, Chemical and Biological Sciences, 3(4):1231-1241.
Lawrencea MJ, Reesb GD, 2000. Microemulsion-based media as novel drug delivery systems. Advanced Drug Delivery Reviews, 45:89–121.
Madhav NVS, Saini A, 2011. A Review on Niosomes: A Novel Drug Delivery System. International Journal Of Research In Pharmacy And Chemistry, 1(3): 498-511.
Mehta A, Jain N, Grobler A, Bharti V, 2016. Role of novel drug delivery systems in bioavailability enhancement. International Journal of Drug Delivery Technology 6(1): 7-26.
Meghna KS, Pillai MK, Giridas S. 2017. Microsphere a drug delivery system: a review. International Journal of Novel Trend in Pharmaceutical Sciences 4(7):109-118.
Nikam VK, Gudsoorkar VR, Hiremath SN, Dolas RT, Kashid VA. 2012. Microspheres - A Novel Drug Delivery System: An Overview. International Journal of Pharmaceutical and Chemical Sciences, 1(1):113-128.
Parimoo P, et al ‘A Text Book of Pharmaceutical Analysis’, CBS publishers and distributors, New Delhi, 1998, 14.
Prabhakar C. 2011. A Review on Nanosuspension in Drug Delivery. International Journal of Pharma and Bio Sciences, 1(2):549-558.
Patil S, Mhaiskar A, Mundhada D. 2016. A Review On Novel Drug Delivery System: A Recent Trend. International Journal of Current Pharmaceutical & Clinical Research, 6(2):89-93.
Prathap B, Dey A, Rao GS, 2013, A Review on Impurity Profile in Pharmaceutical Substances. Journal of Pharmacy and Pharmaceutical Sciences, 2(3):2320-1215.
Panyam J, Labhasetwar V. 2003. Biodegradable nanoparticles for drug and gene delivery to cells and tissue. Advanced Drug Delivery Reviews 55:329–347.
Rangasamy M, Kugalur GP. 2010. Recent advances in novel drug delivery System. International Journal of Research in Ayurveda & Pharmacy, 1(2):316-326.
Sung JC, Pulliam BL, Edwards DA. 2007. Nanoparticles for drug delivery to the lungs. TRENDS in Biotechnology, 25(12)
Srinivas BR, 2015. Formulation and in vitro evaluation of liposome loaded with Mupirocin. International Journal of Research in Pharmaceutical and Nano Sciences, 4(3):162-174.
Sharma A, Sharma S. 1997. A Review on liposome in drug delivery: progress and limitation, International Journal of Pharmaceutics 154: 123-140.
Singh S. 2000. Stability testing during product development in Jain NK, Pharmaceutical product development CBS publisher and distributors. India, pp 272-293.
Tangri P, Bisht B. 2012. Who Role And Guidelines in Stability Study of Pharmaceuticals: A Regulatory Perspective. International Journal of Research in Pharmaceutical and Biomedical Science, (3)3.
Tiwari G, Tiwari R, Sriwastawa B, Bhati L, Pandey S, Pandey P, Bannerjee SK, 2012. Drug delivery systems: An updated review. International Journal of Pharmaceutical Investigation, 1(2).
Thakkar H, Sharma RK, Mishra AK, Chuttani K, Murthy RSR. 2004. Efficacy of Chitosan microspheres for Controlled Intra-articular Delivery of Celecoxib in Inflamed Joints. Journal of Pharmacy and Pharmacology, 56(4):1091-1099.
Witharana C, Wanigasekara J. 2017. Drug Delivery Systems: A New Frontier in Nano-technology. International Journal of Medical Research and Health Sciences, 6(9): 11 – 14.
Yadav GV, 2012. Nanosuspension: A Promising Drug Delivery System. Pharmacophore (An International Research Journal), 3(5):217-243.