

Rameshwar Dass1, D. C. Bhatt2, S. K. Singh2
1Guru Gobind Singh College of Pharmacy, Yamunanagar-135001, Haryana, India
2Guru Jambheshwar University of Science and Technology, Hisar-125001, Haryana, India
*Corresponding author
Rameshwar Dass
Guru Gobind Singh College of Pharmacy,
City Center Road, Yamuna Nagar-135001, Haryana, India
Phone No. 09896503552, Fax-01732-200381
Abstract
Objective: In the current research work, chitosan, bovine serum albumin and sodium alginate microspheres containing metoclopramide hydrochloride were prepared and evaluated for in-vitro and ex-vivo performance of metoclopramide hydrochloride. Materials and methods: Microspheres containing metoclopramide hydrochlorides were prepared by emulsification solvent evaporation method. The surface morphological characteristics of different microspheres were investigated using microscopy. The polymer ratio, stirring speed and the temperature affected the particle size, shape and surface morphology of the microspheres. The in-vitro drug release was carried out using USP paddle type dissolution rate test apparatus phosphate buffer (pH 7.4) dissolution medium at 272nm. Results and conclusion: The best entrapment was achieved in chitosan microspheres i.e. 91.95%. By, above results it was concluded that chitosan microspheres showed reproducible results and with good surface morphology. The above study results that metoclopramide hydrochloride microspheres appropriate system for nasal delivery.
Keywords: Metoclopramide hydrochloride, chitosan, bovine serum albumin, sodium alginate, nasal delivery
Introduction
Nasal delivery is considered to be a promising technique for the subsequent reasons: the nose has a large surface area available for drug absorption due to the coverage of the epithelial surface by numerous microvilli, the subepithelial layer is highly vascularized, the venous blood from the nose passes directly into the systemic circulation and therefore avoids the loss of drug by first-pass metabolism in the liver, it offers lower doses, more rapid attainment of therapeutic blood levels, quicker onset of pharmacological activity, fewer side effects, high total blood flow per cm porous endothelial basement membrane, it is easily accessible, and drug is delivered directly to the brain along the olfactory nerves (Illum et al., 1994; Kissel et al., 1998; Ridley et al., 1995).
Microspheres carriers prepared with numerous polymers are attracting attention for many years for sustained release. Current scenario of dosage form is managed release rate and site specific to develop new drug delivery system. Microspheres are part of this type of novel development of system (Woo et al., 2001; Capan et al., 2003; Gogel et al., 1998).
The achievement in this system is low due to shorter stay at absorption site and rare absorption of large molecules. This limitation is over come by blend of bioadhesion properties to microspheres. Such microspheres improve the residence time and intimate contact with absorption site (Nagai et al., 1984; Ilium et al., 1988; Schaefer et al., 2000; Swamy et al., 2012).
The limitation of nasal drug delivery is the mucociliary clearance that provides a limited available for asorption within the nasal cavity (Casettari et al., 2014). Chitosan and serum albumin were employed as matrix where drug was dispersed in microspheres. These polymers are chosen due to their mucoadhesive properties to mucosal layer (Goncalves et al., 2005).
Material and methods
Drug and chemicals
The drug Metoclopramide hydrochloride was obtained as gift sample from Ind-Swift Ltd. Haryana, India. The mucoadhesive polymers used were chitosan (CIFT, Kochin, India), Bovine serum albumin (Sisco Research Lab., Mumbai, India) and sodium alginate (Otto Kemi, Mumbai). All the other chemicals used in the study were purchased from S.D. Fine Chemicals, Mumbai, India and Qualigens Fine chemicals, Mumbai, India.
General method for preparation of microspheres
Bovine serum albumin microspheres
Drug loaded microspheres were prepared by emulsion solvent evaporation method. Bovine serum albumin (24% w/v) was dispersed in phosphate buffer pH 7.4. To this solution, (1:2, drug: polymer) Metoclopramide hydrochloride was added. The above bovine serum albumin dispersion was added to the continuous phase (10ml) which was a mixture of liquid paraffin and petroleum ether (6:4 v/v). The two phases were mixed in a vortex shaker for 5 min, to obtain a water-in-oil emulsion. Cross-linking of the microspheres was carried out by adding 1-2 ml of 25% glutaraldehyde and further stirring the dispersion for 20 min at room temperature (Bregnic et al., 2000).
Chitosan microspheres
The microspheres were prepared by the suspension cross linking method. Chitosan (about 200 mg) was dissolved in 10 ml 5% acetic acid solution and this solution (1:5 drug: polymer) dispersed in 60 ml of the suspension medium containing mineral oil/petroleum ether (60/40, ml) mixture and 1ml of span 80 as an emulsifier. The suspension medium was stirred (Remi stirrer, Mumbai, India) at constant speed for 10 minutes and 1ml of glutaraldehyde was added into the medium and moving persistent. With subsequent addition of 0.5 ml of glutaraldehyde after 30 min into the suspension medium, stirring were continued. At the end of 1 hour, stirring was stopped and the Chitosan microspheres were washed several times with petroleum ether, 5% sodium bisulphate solution and acetone, (consecutively) and then dried in an oven 65 °C (Bregnic et al., 2000).
Sodium alginate microspheres
The microspheres were prepared by cross linking of sodium alginate, participating in the aqueous internal phase of the primary emulsion with calcium ions (CaCl2 solution 10 % w/w). The supernatant is decanted, the sediment was washed with distilled water (200mL) 3 times and the microspheres was filtered and dried at 37 ºC, until they reached a constant weight12. The external oil phase, containing 70mL of liquid vaseline, 30mL of petroleum ether, and span 80 (3%; w/w) was emulsified in an equal volume of sodium alginate solution, by means of a small vortex shaker to produce the primary water in oil emulsion (Thanoo et al., 1992).
Characterization of prepared microspheres
Particle size analysis
The particle size and shape of the microspheres of different polymers was determined by optical microscopy. Two hundred microspheres were counted using a calibrated optical microscope. The sample microspheres were placed on slide and then observed under the calibrated optical microscope (Liu et al., 1997).
Shape and surface morphology
The surface of microspheres was evaluated via binocular microscope. The sample was mounted on to glass slide and covered with cover slip (Liu et al., 1997).
Incorporation efficiency
The incorporation efficiency was determined by the method of briefly, 50 mg of drug-loaded microspheres were digested in 10 to 25ml of 7.4pH buffer solution for 24 hr at room temperature (25 °C) on magnetic stirrer to release any entrapped drug. The supernatant layer was removed and diluted properly and measured the actual drug content by analyzing the solution by UV Spectrophotometer (UV-1800 SHIMAZU) at 272nm.
% Entrapment efficiency = (Actual drug content/ theoretical drug content) ×100.
In-vitro release studies
In vitro drug release studies were performed via dialysis technique. Dialysis bags having suspension of drug loaded microspheres (10mg drug) in 5ml buffer solution of pH7.4 were placed in 250ml of phosphate buffer medium at 37±0.5oC and stirred at 50rpm using USP paddle method. The samples were assayed for drug release via UV spectrometer at 272nm and then calculate cumulative release of formulation (Lehr et al., 1992).
Ex- vivo nasal perfusion studies
The rats (200-225 g) were anaesthetized by intraperitoneal injection of pentobarbitone sodium (30-50 mg/kg body weight). After an incision is made in the neck, trachea was cannulated with a polyethylene tube. The passage of the nasopalatine tract was sealed to prevent drainage of drug solution from nasal cavity through the nostril or through the esophagus cannulation tubing. During perfusion, a funnel was placed between the nasal cavity and reservoir to minimize loss of drug solution. The drug solution/suspension of microspheres, which was held in a reservoir maintained at 37±0.5oC, was circulated through the nasal cavity of the rat by means of a peristaltic pump. The perfusion solution/suspension of microspheres passed out of the nostrils, through the funnel, and returned to the reservoir, where the solution was stirred constantly with a magnetic stirrer. The amount of drug absorbed was determined by measuring the initial and final drug concentration of the perfusing solution. When all possible outlets in the rat nasal region were blocked following surgical preparation, the only possible passage for the drug to be absorbed and transported into the systemic circulation was by penetration and/ or diffusion through the nasal mucosa (Illum et al., 1994; Patel et al., 2004).
Statistical data analysis
Results of ex- vivo experiments are reported as SEM (n= 5). Statistical tests of significance were performed using (Graph Pad in stat 3 software), differences were considered to be statistical significant when p< 0.05 using a two- tailed unpaired test (Deasy et al., 1994; Gavini et al., 2006).
Results and discussion
Albumin, chitosan and sodium alginate microspheres were prepared by emulsion solvent evaporation method. The particle size of microspheres was determined by optical microscopy. The average particle size was found to be in range of batches A3, C4 and SA6, 10 to 60µ, 1-20µ and 1-50µ respectively. Chitosan polymer showed spherical nature of microspheres as illustrated in figure. It was studied with escalating the concentration of polymers particle size improves. Above mean particle size of different polymers was found with at concentration of 24%, 5% and 2%. The particle size of microspheres above and lower than optimum concentration demonstrates coalescence of droplets. The preparation of different polymers microspheres occupied with upholding at 20 to 25oC.
Optical microscopy of selected batches
Shape and surface morphology were considered with microscopy. Pictures of different polymers were indicated showed nearly spherical and smooth surface (Figure 1). On the basis of particle size distribution the selected batches (form six batches of each polymer) of albumin, chitosan and alginate microspheres is depicted in table 1.
Table 1. Selected batches (form six batches of each polymer) of albumin, chitosan and alginate microspheres
|
Batch No. |
Polymer solution (w/v) |
Polymer: pet. Ether ratio |
Drug: polymer ratio |
%age of Emulsifying agent (w/w) |
Cross linking agent for each 10ml |
Time of stirring (minutes) |
|
A2 |
24% |
7:3 |
1:2 |
0.4 |
2 |
60 |
|
A3 |
24% |
7:3 |
1:2 |
0.4 |
4 |
30 |
|
C2 |
2% |
6:4 |
1:5 |
0.4 |
1 |
60 |
|
C4 |
2% |
6:4 |
1:5 |
0.4 |
2 |
60 |
|
S.A. 5. |
4% |
6:4 |
1:5 |
0.3% |
10 |
20 |
|
S.A. 6. |
5% |
6:4 |
1:5 |
0.3% |
10 |
20 |
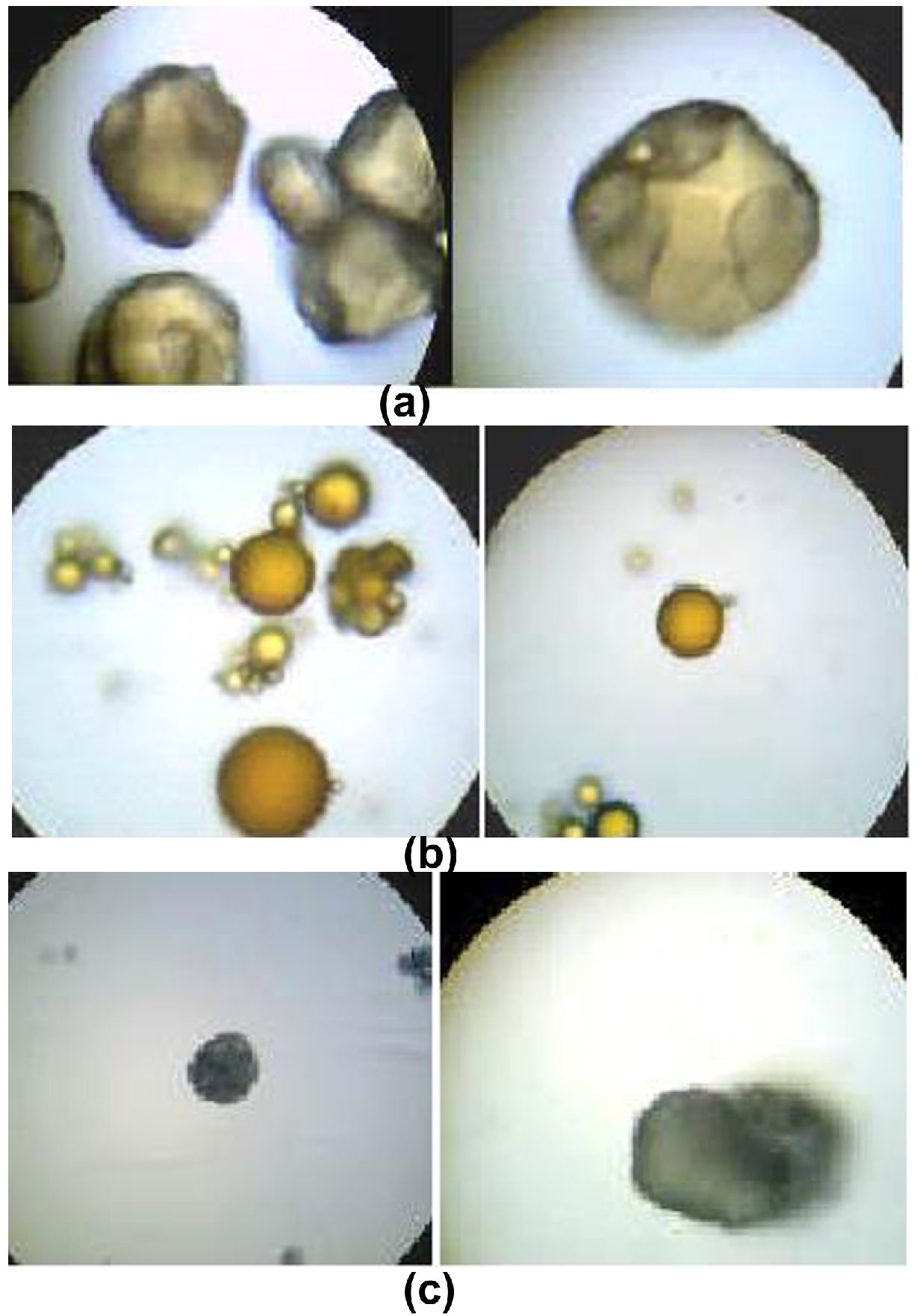
Figure 1. (a) Albumin microspheres (b) Chitosan microspheres and (c) Sodium Alginate microspheres
Mucoadhesive properties
The microspheres were evaluated for mucoadhesive activity by In-vitro wash off test. Adhesion property was evaluated through USP/IP/BP disintegration apparatus wherein container filled with 1.2pH buffer solution. The microspheres sticking to the tissue were measured in form of numbers of particles stand by after 30 minutes, 1 hour and at 4 hr. the calculation show that batch A3, C4 and SA6 presented maximum 75% mucoadhesion as compare to other batches (Table 2). In- vitro wash off test among percentage of mucoadhesion and time in hours of A3, C4 and SA6 batches is shown in figure 2.
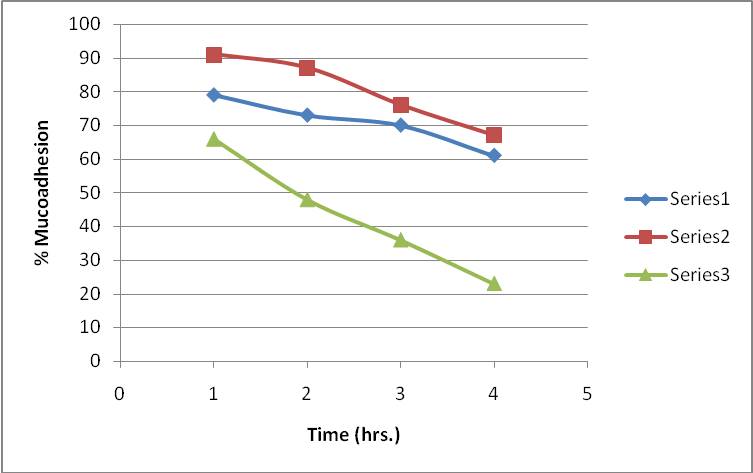
Figure 2. In-vitro Wash off test
Table 2. Result of In-vitro mucoadhesive properties of the microparticles
|
Batch Code |
% Mucoadhesion to Stomach Mucosa |
|||
|
After 1 HR |
After 2 HR |
After 3 HR |
After 4 HR |
|
|
A3 |
79 |
73 |
70 |
67 |
|
C4 |
91 |
87 |
76 |
74 |
|
SA6 |
66 |
48 |
36 |
23 |
Incorporation efficiency
50 mg of drug-loaded Microspheres were digested in 10 to 25ml of 7.4pH buffer solution for 24 hrs. at room temperature (25 °C) to release any entrapped drug. The supernatant layer was removed and diluted 10 times. The actual drug content was found by Spectrophotometry at 272nm. Incorporation efficiency was calculated using the following formula
% entrapped drug = (Actual drug content/ theoretical drug content) ×100 (Table 3).
Table 3. Incorporation efficiency of drug in different batches
|
Batch Code |
Drug: polymer ratio |
% Drug content |
|
A3 |
1:2 |
66.55+ 0.003 |
|
C4 |
1:5 |
75.94+ 0.002 |
|
SA6 |
1:5 |
63.05+ 0.003 |
In vitro release studies
The dissolution studies of selected batches of microspheres were carried out in 250 ml phosphate buffer of pH 7.4, rpm 50 at 272nm. The maximum amount of drug released in 6 hours was found to be 95, 98.1 and 88 of batches A3, C4 and SA6 (Table 4).
Table 4. In-vitro drug release study of three batches
|
Time (minutes) |
% Cumulative release content |
||
|
A3 |
C4 |
SA6 |
|
|
10 |
3.6 |
2.68 |
4.04 |
|
15 |
7.83 |
7.38 |
8.93 |
|
30 |
13.38 |
12.61 |
14.54 |
|
60 |
20.91 |
19.25 |
21.35 |
|
90 |
29.47 |
26.62 |
29.93 |
|
120 |
39.12 |
35.03 |
39.44 |
|
180 |
50.14 |
48.95 |
49.87 |
|
240 |
63.7 |
64.17 |
61.43 |
|
300 |
78.62 |
80.96 |
74.11 |
|
360 |
95.19 |
98.1 |
87.76 |
% Cumulative release of drug from batches A3, C4 and SA6 is given in figure 3.
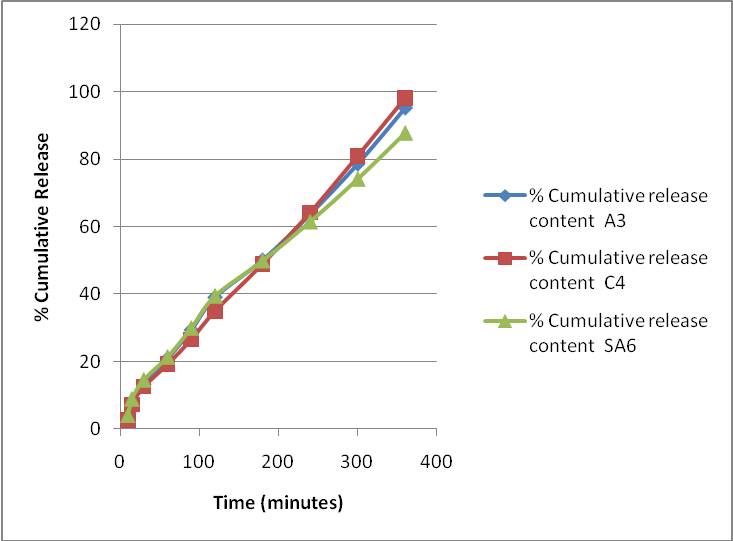
Figure 3. In-vitro release studies
Stability Studies
![]()
![]()
![]() A stability study of drug was done for different batches and was stable after 3 months of degradation studies of metoclopramide hydrochloride and maintained for period.
A stability study of drug was done for different batches and was stable after 3 months of degradation studies of metoclopramide hydrochloride and maintained for period.
Conclusion
The best entrapment was achieved in chitosan microspheres i.e. 91.95%. By, above results it was concluded that chitosan microspheres showed reproducible results and with good surface morphology. The above study results that metoclopramide hydrochloride microspheres appropriate system for nasal delivery.
Conflicts of interest: Nil
References
Bregnic, Degnorsi T, Garcia R, Lamn MC, Firenstein R, D'Aquino M. 2000. Alginate microspheres of Bacillus subtilis. Ars Pharmaceutica, 41:3; 245-248.
Capan Y, Jiang G, Giovagnoli S, De Luca PP. 2003. Prepararion and characterization of poly (D, L-lactide-co-glyclide) microspheres for controlled release of human growth hormone. American Association of Pharmaceutical Scientists Pharmaceutical Science and Technology, 4:E28.
Casettari L, Illum L. 2014. Chitosan in nasal delivery systems for therapeutic drugs. Journal of Control Release, 190, 189-200.
Deasy PB. 1984. Microencapsulation and Related Drug Processes. Drugs and pharmaceutical sciences series, vol. 20. pp.61–96. Marcel Dekker, New York.
Gavini E, Hegge AB, Rassu G, Sanna V, Testa C, Pirisino G, Karlsen J, Giunchedi P. 2006. Nasal administration of carbamazepine using chitosan microspheres: In vitro/in vivo studies. International Journal of Pharmaceutics, 307:9-15.
Gogel MC,Amin AF. 1998. Formulation optimization of controlled release diclofenac sodium microspheres using factorial design. Journal of Control Release, 51:115-122.
Goncalves, VL, Laranjeira MCM, Favere VT. 2005. Effect of crosslinking agents on chitosan microspheres in controlled release of diclofenac sodium. Polymers Science Technology, 15:6-12.
Ilium L, Farraj NF, Critchley H, Davis SS. 1988. Nasal administration of gentamicin using a novel microsphere delivery system. International Journal of Pharmaceutics, 46:261-265.
Illum L, Farraj NF, Davis SS. 1994. Chitosan as novel drug delivery system for peptide drugs. Pharmaceutical Research, 11:1186-1189.
Kissel T, Werner U, 1998. Nasal delivery of peptides: an in vitro cell culture model for the investigation of transport and metabolism in human nasal epithelium. Journal of Control Release, 53:195–203.
Lehr CM, Bouwstra JA, Schacht EH, Junginer HE. 1992. In vitro evaluation of Mucoadhesive properties of chitosan and some other natural polymers. International Journal of Pharmaceutics, 78:43-48.
Liu LS, Liu SQ, Ng SY, Froix M, Heller J. 1997. Controlled release of interleukin 2 for tumour immunotherapy using alginate/chitosan porous microspheres. Journal of Control Release, 47:65-74.
Nagai T, Nishimoto Y, Nambu N, Suzuki Y, Sekine K. 1984. Powder dosage form of insulin for nasal administration. Journal of Control Release, 1:15-22.
Patel JK, Bodar MS, Amin AF, Patel MM. 2004. Formulation and optimization of Mucoadhesive microspheres of metoclopramide. Indian Journal of Pharmaceutical Sciences, 66:300-305.
Ridley D, Perkins AC, Washington N, Wilson CG, Wastie ML, Flynn PO, Blattman A, Ponchal G, Duchene D. 1995. The effect of posture on nasal clearance of bioadhesive starch microspheres. SonaTeb Parseh Pharmaceutical Science, 5:442–6.
Schaefer MJ, Singh J. 2000. Effect of isopropyl myristic acid ester on the physical characteristics and in vitro release of etoposide from PLGA microspheres. American Association of Pharmaceutical Scientists Pharmaceutical Science and Technology, 1:E32.
Swamy NGN, Abbas Z. 2012. Preparation and in vitro charactrisation of mucoadhesive polyvinyl alcohol microspheres containing amlodipine besylate for nasal administration. Indian Journal of Pharmaceutical Education and Research, 46:52-58.
Thanoo BC, Sunny MC, Jayakrishnan A. 1992. Cross-linked chitosan microspheres: preparation and evaluation as a matrix for the controlled release of pharmaceuticals. Journal of Pharmacy Pharmacology, 44:283-286.
Woo BH, Jiang G, De Luca PP. 2001. Preparation and characterization of the composite PLGA and poly (acryloyl hydroxymethyl starch) microsphere system for protein delivery. Pharmaceutical Research, 18:1600-1606.