

Antoine Kada Sanda1*, David Fotsing1, Oumar Mahamat1, Vijayakumar Natesan2, Jayaprakash Rajendran3, Edouard Akono Nantia4, Paul Desire Dzeufiet Djomeni5, Pierre Kamtchouing5
1Department of Biological Sciences, Faculty of Science, University of Bamenda, POBox 39, Bambili, Cameroon
2Department of Biochemistry and Biotechnology, Faculty of Science, Annamalai University, Chidambaram-608002, Tamilnadu, India
3Department of Chemistry, School Of Arts And Science, AV Campus, VMRF, Paiyanoor, Tamilnadu, India
4Department of Biochemistry, Faculty of Science, University of Bamenda, PoBox 39, Bambili, Cameroon
5Department of Animal biology and physiology, faculty of science, university of Yaoundé 1, PoBox 812, Yaoundé, Cameroon
* Corresponding Author:
Kada Sanda Antoine
Department of Biological Sciences
University of Bamenda, PO BOX: 39, Bambili, Cameroon
Abstract
Objective: Covid-19 like pandemics is encouraging the plant based medicines nowadays. The Allanblackia floribunda (Clusiaceae) stem bark extract is used to treat the sexual dysfunctions problems in Cameroon traditionally. The reported oral studies research in adult male rats using the bark extract for eight days revealed the increased frequencies of erection, intromission and prolonged latency of ejaculation, while intravenous and oral administration delayed the occurrence of ejaculation in spinal male rats. The present study aimed to investigating the androgenic, androgen releasing property of aqueous extract of A. floribunda stem bark and its relation with corporal nitric oxide (NO) production in male albino adult Wistar rats. Materials and methods: This extended work was done to investigate the androgenic and androgen releasing tendency of A. floribunda stem bark aqueous extract in male albino Wistar rats through assessment of the corporal nitric oxide, testosterone, LH production, reproductive organs weight and accessory reproductive organs secretions. Results: Treatment of animals with A. floribunda extract at the dose 150 mg/kg increased serum testosterone and luteinizing hormone. In testis homogenate, it increased testosterone and cholesterol levels, while enhancing penile nitrites and vesicular fructose. Moreover, animals treated with A. floribunda extract displayed high red blood cells number, hematocrit level, thickness and diameter of the seminiferous tubules. Conclusion: A. floribunda improves the male reproductive of male rats through its androgenic properties and improvement of biomarkers of accessory reproductive organs.
Keywords: Allanblackia floribunda; testosterone, androgenic properties, nitric oxide, male rats
Introduction
The sex hormones androgens are responsible of the devolvement of male characters in men. The presence of enough androgens (especially testosterone and its metabolite dihydrotestosterone) is promoting embryonic differentiation of the fetus into a male and further growth in the male reproductive organs and body muscle mass (Baskin et al., 1997). Another type of androgens, dehydroepiandrosterone secreted from the adrenal glands also influences the sexual characteristics of the men (King et al., 2002). Androgen deficiency (hypogonadism) at the early developmental phase leads to the structural abnormalities of the penis (Mills et al., 1996; Shabsigh, 1997). Hypogonadism in adulthood may lead changes like loss of libido and sexual activity respectively as well as atrophy of the reproductive organs. Androgens are responsible for sex drive in males and their involvement has also been demonstrated in the erection process (Meisel and Sachs, 2001). Many studies using rats demonstrated previously that sexual behaviour and erection are androgen dependent (acting both centrally and peripherally) and that treatment with testosterone of hypogonadic men helped in restoration of both sexual behaviour and penile erectile capacity (Burris et al., 1992; Carani et al., 1996; Mills and Lewis, 1999). It has also been demonstrated by various studies that androgens induced penis erection by regulating corporal nitric oxide synthase (NOS) activity or by influencing cyclic guanosine monophosphate (cGMP) level through direct action on type 5 phosphodiesterase (PDE5) (Zvara et al., 1995; Reilly et al., 1997). Populations in Cameroon (Central Africa) are using Allanblackia floribunda to treat traditionally sexual weakness and male infertility. Previous studies of the pharmacological activities of A. floribunda extracts have demonstrated the aphrodisiac properties of his stem bark extracts and some fractions isolated from the plant (Kada et al., 2012; Kada et al ., 2013; Brusotti et al., 2016). Eight days oral administration of the A. floribunda stem bark aqueous extract increased the frequencies of erection (mount), intromission and prolonged latency of ejaculation in adult male rats. Aqueous and ethanolic extract of A. floribunda delayed the ejaculation induced by dopamine in spinal male rats. The chemical constituents of crude ethanolic extract such as fukugiside, spicataside and morelloflavone were identified in the different fraction of A. floribunda extract. Aphrodisiac substances act by increasing testosterone production in male rats or by acting as agonists of androgen receptors (Sandroni, 2001). From our previous findings who demonstrated that A floribunda extracts possess aphrodisiacs activity (Kada et al., 2012; Kada et al., 2013; Brusotti et al., 2016), we hypothesized that this aphrodisiac effect of the plant can be due to it androgenic potential. The present study aimed to investigating the androgenic, androgen releasing property of aqueous extract of A. floribunda stem bark and its relation with corporal nitric oxide (NO) production in male albino adult Wistar rats.
Material and methods
Chemicals, drugs and assay kits
Double distilled water was prepared in our laboratory for the extraction. Testosterone undecanoate was purchased from Merck Sharp & Dohme, Australia, Ethylene Diamine Tetra Acetic Acid (EDTA) from Lab Chem Inc, Jackson’s Pointe Court Zelienople, USA. Remaining chemical reagents are analytical grade reagents and were supplied by Sigma Chemicals, St. Louis, USA. The kits for cholesterol, testosterone and luteinizing hormone were obtained from Roche Diagnostic, Mannheim, Germany. Commercial kit (ELYSA) manufactured by Human Gesellschaftfür Biochemica and Diagnostic ambH.
Collection, dressing and extraction of Allanblackia floribunda
Fresh stem bark of A. floribunda was collected from the Okala situated in the Center region of Cameroon for the study. The plant was confirmed by R. Letouzey in the Cameroon National Herbarium (CNH) and a voucher specimen (1380/HNC) was deposited for the further work. The dressed stem bark of the plant was cleaned, slashed into pieces and dried at the room temperature for a week. The dried bark was crushed well and 100 g of the obtained powder were macerated in 1 L of distilled water at room temperature for 1 day, and the mixture filtered using Watmann-3 filter paper. The filtrate was evaporated at 45°C under vacuum 3.68 g of brown dried extract were obtained.
Phytochemical analysis
Different spot test reagents such as Mayer’s test, lead acetate test, Salkowski’s test and Ferric chloride test were used to identify the natural phytochemical constituents of Stem bark of A. floribunda and the outcomes were measured based on the intensity of the results (Ackah et al., 2021).
Animals
Healthy adult male Wistar rats of the species Rattus norvegicus (weighing 240 to 250 g) of 2.5 months old were purchased from the Faculty of Science, University of Yaoundé 1 animal house. They were kept in standard conditions in clean cages with enough spaces and were subjected to the natural light-dark cycles and provided free access to have food and water. All the experiments were conducted with the authorization of the Cameroonian national ethical committee of the use of laboratory animals (Ref No. FW-IRB00001954).
Animal grouping and treatment
Thirty two rats were grouped into four sets of eight rats each randomly constituted. The animals of were orally administered 10 mL/kg distilled water (negative control group), 2.6 mg/kg of the testosterone undecanoate (positive control group) or 150 mg/kg and 300 mg/kg of the aqueous extracts of A. floribunda for 8 consecutive days. The doses of the aqueous extracts of A. floribunda were selected from previous studies on the aphrodisiac activity this plant (Kada et al., 2012; Kada et al., 2013; Brusotti et al., 2016) , while the dose of testosterone undecanoate was calculated from the dose prescribed in hypogonadism affected men.
Preparation of serum and sexual organs homogenates
After eight days treatment, the rats under ether anesthesia were sacrificed early in the morning (in order to reduce the influence on hormonal changes due to circadian rhythm), and jugular venous blood was collected into dry tubes and tubes with EDTA (Ethylene Diamine Tetra Aceticacid) anticoagulant. Plasma obtained from blood with anticoagulant was used for hematological analyzes (red blood cells, hemoglobin and hematocrit), meanwhile serum was used for analysis of hormonal assay and biochemical tests. The testes, epididymis, prostate, vas deferens, seminal vesicle, penis and an elevator muscle were dissected and weighed. The right testis and the right epididymis were fixed in Bouin for histological studies . The homogenates from penis, prostate, seminal vesicle, left testis and epididymis were for biochemical analyses.
Biochemical analyses
Total proteins in serum and reproductive organ (testis and epididymis) homogenates were determined using colorimetric methods method of Gornal et al (1949) and Bradford, (1976) respectively . Fructose levels were estimated in seminal vesicle using WHO protocols (OMS, 1993). Commercial kit was used to determine the level of cholesterol in testis and serum with the according to the manufacturer instructions. Nitrite contents of the penis were determined using method described by Ikeda et al, (2003).The prostatic acid phosphatase activity was quantified according to the method described by Lin and Clinton in (1986). Blood parameters (red blood cells, hemoglobin and hematocrit) were assessed using and automatic counter branded Hycel Diagnostics Diana-5 Evolution, version V5.34 N/5 E.568.
Histological and histomorphomotry analysis
The right testis and epididymis fixed in the Bouin solution were then embedded in paraffin and the sections of 4–5 µm were prepared and stained with haematoxylin and eosin (H&E). The sections were examined through microscope. Morphometric parameters such as surface, diameter and densitometry on histological sections of an organ were measured to identify the dose effect of the treatment on the histological structure of the rat testes. They were measure using OLYMPUS microscope and the image was capture by a Compaq nx9010 computerized DCM35 350K pixels digital camera. Data were recorded on 4 glass microscope slide of each group. The average area of orthogonal sections of seminiferous tubules was determined on 10 sections per testes using a built-capture program (Minisee) and area measurement (Image J version 1.32j) under X100 magnification. The Feret's diameter was measured from each other on the periphery of the area.
Statistical analysis
The results were expressed as mean + Standard Error of Mean (SEM). For each animal group, the mean values were calculated by one way ANOVA (Analysis Of Variance). The quantitative data comparisons was analyzed using Student– Newman–Keuls’s posttest using Graph pad Instant software version 3.10 (La Jolla, CA).
Results
Phytochemical compounds in A. floribunda extract
The phytochemical results (Table 1) showed the presence of the various phytocontituents in plant including flavonoids, alkaloids, triterpenoids, xanthones, anthroquinones, glycosides, steroids.
Table 1. Reagents and the Observations of Phytochemical Analysis
|
Phtochemicals |
Test name/method |
Observation |
Intensity |
|
Alkaloids |
Mayer’s test |
yellow |
+ |
|
Wagner’s test |
Reddish brown |
+ |
|
|
Flavonoids |
Lead acetate test |
Yellow precipitate |
++ |
|
H2SO4 test |
Orange colour |
++ |
|
|
Triterpenoids |
Salkowski’s test |
Red brown |
++ |
|
Xanthones / Anthroquinones |
Borntrager’s test &TLC (7:3 chloroform ðyl acetate), NH3 spray in UV |
Light pink colour |
+ |
|
Glycosides |
NaOH + 2 % solution of 3,5- dinitro benzoic acid |
Pink colour |
+++ |
|
Phenolic compounds |
Ferric chloride test |
Bluish colour |
++ |
|
Carbohydrates(Reducing sugars) |
Benedict’s test |
Orange red precipitate |
++ |
|
Saponins |
Distilled water shaking |
Creamy and bubbles |
+ |
|
Steroids |
Acetic Anhydride+H2SO4 |
Violet to blue |
+ |
|
Oil |
Sprayed on Filter paper |
Transparent |
+ |
Note: +++ - more intensity, ++- moderate, + -less or traces
Effect of A. floribunda stem bark aqueous extract on animal body and organs weight
The dose 150 mg/kg of A. floribunda extract significantly increased (P <0.05) the body weight of the animals when compared to the distilled water treated rats (Table 2). Administration of Testosterone undecanoate highly increased animal weight by (19.40%) when compared to the negative control (11.20%) group. On organ weights, testosterone and 150 mg/kg of the extract increased the testis (82.55%) and epididymis (55.31%) weights when compared to the negative control. The higher dose (300 mg/kg) of the plant extract did not affect the animal body and organ weight weights.
Table 2. Effect of the aqueous extract of A. floribunda on the weight gain and the weight of reproductive organs.
|
Parameters/Treatements |
Distilled water (10mL/kg) |
Testosterone Undecanoate (2.6 mg/kg) |
A. floribunda (150 mg/kg) |
A. floribunda (300 mg/kg) |
|
Weight gain (%) |
11.20 ± 0.96 |
19.40 ± 0.87aaa |
14.70 ± 0.66ab |
13.70 ± 2.31b |
|
Testes (g) |
1.49 ± 0.16 |
2.99 ± 0.09aaa |
2.72 ± 0.06aaa |
1.87 ± 0.27bc |
|
Epididymis (g) |
0.47 ± 0.01 |
0.90 ± 0.04aaa |
0.73 ± 0.09a |
0.52 ± 0.02bbc |
|
Levatorani (g) |
0.50 ± 0.01 |
0.64 ± 0.02a |
0.67 ± 0.04a |
0.55 ± 0.03 |
|
Ventral prostate (g) |
0.11 ± 0.02 |
0.24 ± 0.01a |
0.11 ± 0,01b |
0.13 ± 0.02b |
|
Penis (g) |
0.16 ± 0.01 |
0.25 ± 0.01aa |
0.22 ± 0.01a |
0.20 ± 0.02a |
|
Vas deferens (g) |
0.0056 ± 0.0002 |
0.0087 ± 0.0004 |
0.0916 ± 0.0003aaa |
0.0065 ± 0.0005cc |
|
Seminalvesicle (g) |
0.41 ± 0.03 |
2.19 ± 0.31aaa |
1.10 ± 0.06aaab |
0.57 ± 0.04bbbccc |
Values in the table represent means ± standard error of means (SEM), n=8 rats per group.
aaaP <0.001 ; aaP < 0.01 ; aP < 0.05 compared to the group treated with distilled water
bbbP <0.001 ; bbP < 0.01 ; bP < 0.05 compared to the group treated with Testosterone Undecanoate
cccP<0.001; ccP<0.01; cP<0.05 compared to the group treated with 150 mg/kg of the aqueous extract.
Effect of A floribunda aqueous extract on some biochemical parameters in the serum and the sexual organs
After the initial weight investigation, this research examined the impact of extract in serum and the sexual organs. The observed results are shown in Table 3. From these results, the extract doses did not influenced the level serum proteins when compared to the negative control group treated with distilled water. But positive control showed a significant increase in serum proteins compared to the negative control group. Like the weight result, 150 mg/kg dose significantly increased the level of the following parameters such as testicular proteins, epididymal proteins, vesicular fructose, prostatic acid phosphatase activity, testicular cholesterol and penile nitrite which was almost approximate like positive control. The higher dose 300 mg/kg of the aqueous extract of A. floribunda showed significant increase in prostatic phosphatase acid activity and serum cholesterol.
Table 3. Effect of the aqueous extract of A. floribunda on certain biochemical parameters
|
Parameters/Treatments |
Distilled water (10mL/kg) |
Testosterone Undecanoate (2,6mg/kg) |
A. floribunda (150 mg/kg) |
A. floribunda (300 mg/kg) |
|
SerumProteins (mg/L) |
92.90 ± 1.09 |
193.10 ± 4.43aaa |
89.86 ± 2.94bbb |
92.64 ± 0.31bbb |
|
Testicular Proteins (μg/g of the organ) |
5.12 ± 0.17 |
7.18 ± 0.42aa |
7.12 ± 0.25aa |
5.29 ± 0.40bbcc |
|
Epididymis proteins (μg/g oh the organ) |
6.86 ± 0.36 |
12.40 ± 0.31aaa |
10.40 ± 0.63aaab |
7.08 ± 0.577bbc |
|
Vesicular fructose (µmol/g of the organ) |
466.36 ± 19.19 |
669.44 ± 43.06aaa |
633.80 ± 21.62aa |
466.56 ± 19.75bbbccc |
|
Prostatic phosphatase acid activity (U/L) |
1.41 ± 0.11 |
4.38 ± 0.37aaa |
2.81 ± 0.22aab |
2.70 ± 0.12aab |
|
SerumCholesterol(mg/dL) |
160.40 ± 1.20 |
156.60 ± 3.35 |
159.80 ± 4.11 |
192.80 ± 3.56aaabbbccc |
|
Testicular cholesterol (μg/g of the organ) |
15.10 ± 0.78 |
20.60 ± 1.24aa |
19.80 ± 0.73aa |
14.20 ± 0.58bbcc |
|
Penile nitrite (μM/mL of the homogenat) |
0.32 ± 0.06 |
0.83 ± 0.05aaa |
0.82 ± 0.06aaa |
0.50 ± 0.08 |
Values in the table represent means ± standard error of means (SEM), n=8 rats per group.
aaaP <0.001; aaP < 0.01 ; aP < 0.05 compared to the group treated with distilled water
bbbP <0.001 ; bbP < 0.01 ; bP < 0.05 compared to the group treated with Testosterone Undecanoate
cccP<0.001; ccP<0.01; cP<0.05 compared to the group treated with 150 mg/kg of the aqueous extract.
Effects of the aqueous extract of A. floribunda on the level of serum and testicular testosterone
Eight days treatment with the aqueous extract of A. floribunda at the doses of 150 and 300 mg/kg resulted in a significant increase (P <0.01 and P <0.05) in the serum testosterone level of about 163.77%, 89.55% respectively when compared with the negative control group (figure 1).
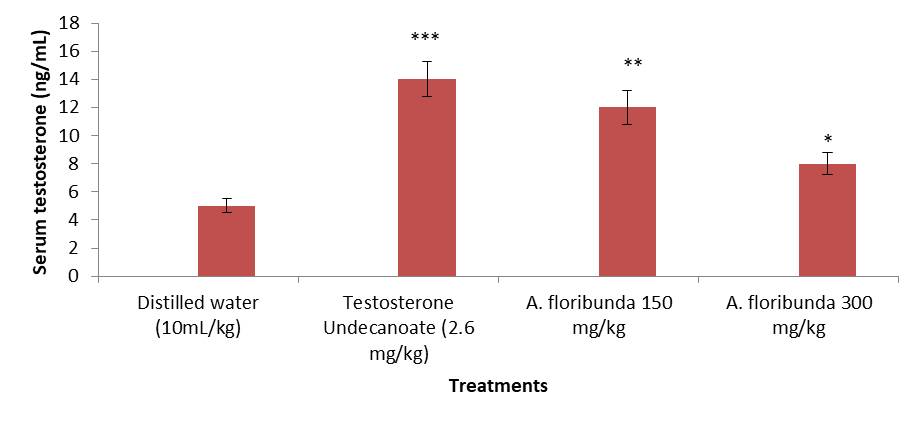
Figure 1. Effect of administration of aqueous extract of Allanblackia floribunda stem on male rat serum testosterone concentration. Each bar represent means ± standard error of means (SEM), n=8 rats per group.***P < 0.001 ; **P < 0.01 ; *P < 0.05 compared to the group treated with distilled water
Figure 2 shows the effect of 8-days treatment with the aqueous extracts of A. floribunda on the testicular testosterone level. From this figure, we have observe that the 150 mg/kg dose exhibited a significant increase (P <0.05) in the level of testicular testosterone when compared to the negative control group.
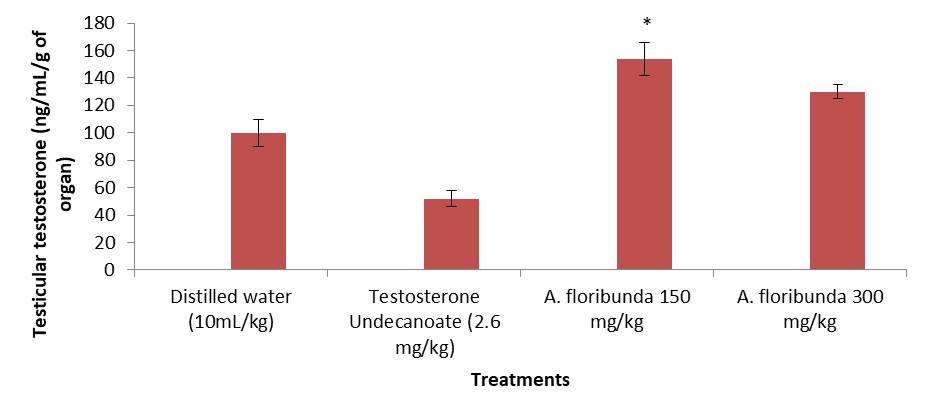 Figure 2. Effect of administration of aqueous extract of Allanblackia floribunda stem on male rat testicular testosterone concentration. Each bar represent means ± standard error of means (SEM), n=8 rats per group. *P < 0.05 compared to the group treated with distilled water
Figure 2. Effect of administration of aqueous extract of Allanblackia floribunda stem on male rat testicular testosterone concentration. Each bar represent means ± standard error of means (SEM), n=8 rats per group. *P < 0.05 compared to the group treated with distilled waterEffect of the aqueous extract of A. floribunda on the level of serum LH
Treatment with the aqueous extract of A. floribunda for 8 days at a dose of 150 mg/kg resulted in a significant increase (P <0.001) in serum LH levels compared to negative control animals (Figure 3). This increase was around 110.52%. On the other hand, testosterone undecanoate at a dose of 2.6 mg/kg caused a significant decrease (P <0.01) in the plasma level of LH compared to the animals of the negative control group.
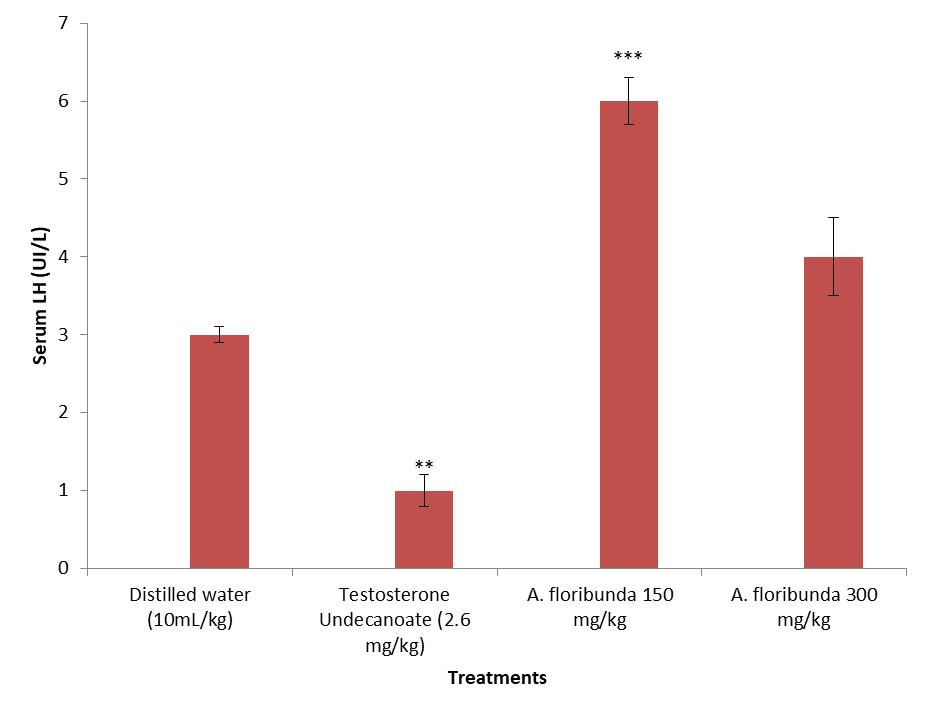 Figure 3.Effect of administration of aqueous extract of Allanblackia floribunda stem on male rat serum LH concentration. Each bar represent means ± standard error of means (SEM), n=8 rats per group.***P < 0.001 ; **P < 0.01 ; compared to the group treated with distilled water
Figure 3.Effect of administration of aqueous extract of Allanblackia floribunda stem on male rat serum LH concentration. Each bar represent means ± standard error of means (SEM), n=8 rats per group.***P < 0.001 ; **P < 0.01 ; compared to the group treated with distilled water
Effect of the aqueous extract of A. floribunda on some blood parameters
Table 4 represents the effects of the aqueous extract of A. floribunda on certain blood parameters. It appears that the 8 days of oral administration of the extract at a dose of 150 m/kg resulted in a significant increase (P <0.01, P <0.01 and P <0.05) in the number of red blood cells, in the levels of hemoglobin and hematocrit respectively compared to the group of animals treated with distilled water. The extract at a dose of 300 mg/kg did not cause any significant variation in these parameters.
Table 4. Effect of aqueous extract of A. floribunda on certain blood parameters
|
Treatments/Parameters |
Red blood cells (10*12/L) |
Hemoglobine (g/dL) |
Hematocrit (%) |
|
Distilled water (10 mL/kg) |
6,58 ± 0,33 |
12,3 ± 0,46 |
34,40 ± 2,56 |
|
Testosterone Undecanoate(2,6 mg/kg) |
9,82 ± 0,51aa |
17,32 ± 0,87aaa |
51,20 ± 2,72aa |
|
A. floribunda 150 mg/kg |
10,20 ± 0,83aa |
15,72 ± 0,51aa |
44,40 ± 2,32a |
|
A. floribunda 300 mg/kg |
5,78 ± 0,42bb |
13,18 ± 3,21bbb |
31,20 ± 3,59bb |
aaaP <0.001; aaP < 0.01 ; aP < 0.05 compared to the group treated with distilled water
bbbP <0.001 ; bbP < 0.01 ; bP < 0.05 compared to the group treated with Testosterone Undecanoate
Effect of the aqueous extract of A. floribunda on histological structure of the testes and epididymis
After the basic studies, the effects of the extract on the sacrificed rat organs were examined by microscopic images (Figure 4 and 5). From the images, the extract doses did not showed significant adverse effects in the structure of the testis and epididymis. Photomicrographs of the testes exhibited a normal structure with germ cells at different stages of spermatogenesis in the seminiferous tubules after the after the eight days of the treatment (figure 4). Likewise, in photographs of the architecture of the epididymis one could perfectly observe the epididymal membrane and their lumen filled with spermatozoa (Figure 5).
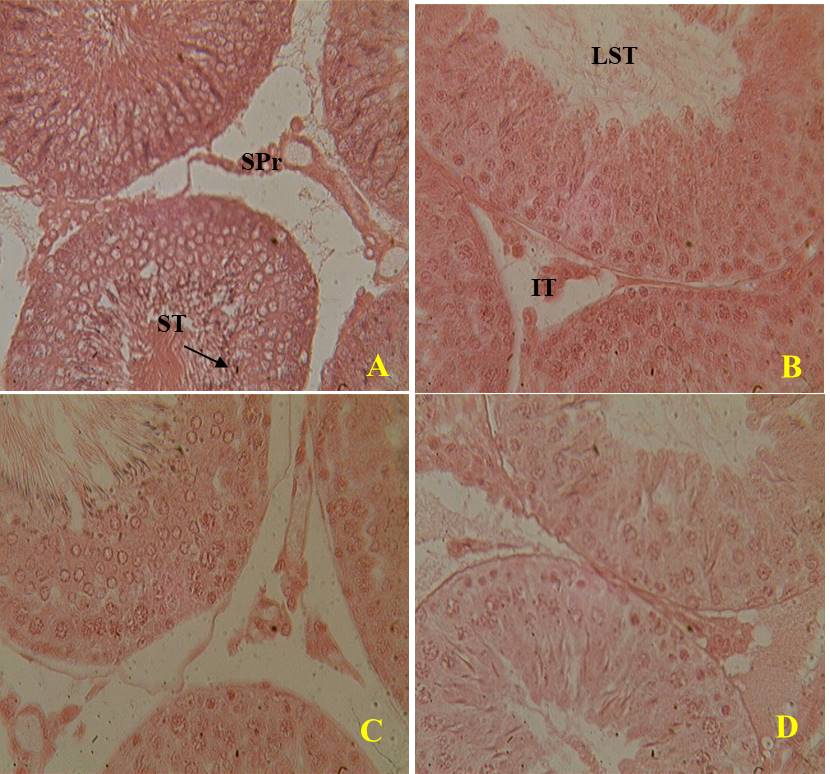
Figure 4. Microphotographs of the testis of rats treated with distilled water (A), Testosterone undecanoate (B), aqueous extract of A. floribunda at doses of 150 mg/kg (C) and 300 mg/kg (D) during 8 days (X400 H&E). (ST) seminiferous tubules, (SPr) sperms, (LST) lumen of the seminiferous tubules, (IT) interstitial tissue.
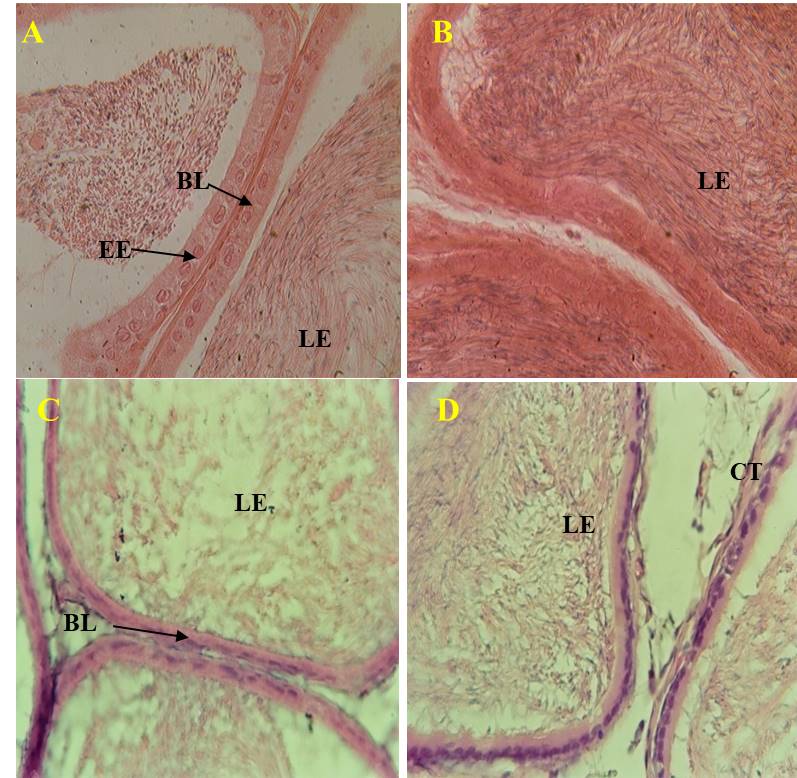
Figure 5. Microphotograhs of the epididymis of rats treated with distilled water (A), Testosterone undecanoate (B), aqueous extract of A. floribunda at doses of 150 mg/kg (C) and 300 mg/kg (D) during 8 days (X400 H&E). (BL) basal lamina, (EE) epithelium of the epididymis, (LE) lumen of the epididymis, (CT) connective tissue.
Effect of the aqueous extract of A. floribunda on histomorphomotry of testis
Table 5 represents the effect of the aqueous extract of A. floribunda on morphometrics parameters of the semineferous tubules. The morphometric study of the testes revealed a significant increase in the thickness of the seminiferous epithelium of approximately 126.15% and 141.53% respectively in animals treated with testosterone and with the aqueous extract at the dose of 150 mg/kg. An increase in the surface area of the seminiferous tubules was observed in animals given the aqueous extract at a dose of 150 mg/kg and testosterone compared to the group of animals given distilled water. However, this increase was only significant (P <0.05) in animals given testosterone. A significant increase (P <0.05) in the diameter of the seminiferous tubules was also observed in the animals treated with the dose of 150 mg/kg of aqueous extract compared to the negative control group.
Table 5. Effect of the aqueous extract of A. floribunda on morphometrics parameters of the seminiferous tubules
|
Treatments/Seminiferous tubules |
Thickness (µm) |
Surface (µm2) |
Diameter (µm) |
|
Distilled water (10 mL/kg) |
0,65 ± 0,06 |
199,12 ± 1,24 |
2,07 ± 0,18 |
|
Testosterone Undecanoate (2,6 mg/kg) |
1,47 ± 0,15aa |
220,82 ± 9,19a |
3,82 ± 0,33aaa |
|
A. floribunda 150 mg/kg |
1,57 ± 0,17aa |
207,20 ± 4,35 |
3,04 ± 0,29a |
|
A. floribunda 300 mg/kg |
0,89 ± 0,12bb |
196,54 ± 1,52b |
1,56 ± 0,22bbbcc |
Values in the table represent means ± standard error of means (SEM), n=8 rats per group.
aaaP <0.001 ; aaP < 0.01 ; aP < 0.05 compared to the group treated with distilled water
bbbP <0.001 ; bbP < 0.01 ; bP < 0.05 compared to the group treated with Testosterone Undecanoate
ccP<0.01compared to the group treated with 150 mg/kg of the aqueous extract
Discussion
Phytochemical screening carried out on the aqueous extract of A. floribunda stem bark has revealed the presence of saponis, alkaloids, flavones and triterpènes. Also, the reported high phase liquid chromatography coupled with electrospray ionization and mass spectrometry (HPLC-ESI-MS) applied to A. floribunda extract led to the isolation and characterization of 4 biflavonoides known as Fukugiside, Morelloflavone, spicataside and volkensiflavone (Brusotti el al., 2016). The androgenic and or anti-androgenic potential of plant extracts can be measured by assessing various parameters such as body weight, biochemical parameters, weight of reproductive organs, body weight ratio, concentrations of testicular secretory constituents (Yakubu et al., 2013; Tetsatsi et al., 2019). After eight days treatment using controls and A. floribunda stem bark aqueous extract doses, the increase in body mass and relative mass of the sex organs were examined successfully and observed with the dose of 150 mg/kg . The increase in the body weight and the weight of reproductive organs is a sign of androgenic potential of the aqueous extract of A. floribunda since reproductive organs weight is an androgen-dependent process (Yakubu et al., 2013, Tetsatsi et al., 2019) Increased functional activity of reproductive organs is express by an increase in their secretory activities and can also explain the increased weigh of the reproductive organs. From the results, we have observed in this work the significant increased of various parameters such as cholesterol levels, testicular protein, testosterone level, level of epididymal proteins, vesicular fructose level, penile nitrite level and prostatic acid phosphatase activity with the aqueous extract at the dose of 150 mg/kg. These results confirmed the presence of bioactive molecules in A. floribunda aqueous extract who have the ability to exhibit androgenic and anabolic effects. Similar results were reported by Mbongue et al., (2010) and Watcho et al., (2004) on their researchs on Aframomum melegueta and Mondia whitei, extracts respectively on rats.
Epididymal and testicular proteins are the major constituents which ensure the maturation of the spermatozoa (Kasturi et al., 1995). Increasing weight, high protein concentration of the epididymis and testes revealed the enhancement of testicular and epididymal growth (Means, 1975). This work also showed the increased levels of testicular protein with the extract which may be attributed to testosterone action (Gupta et al., 2004). Such increase in protein concentration is an indication of anabolizing and androgenic effects of A. floribunda aqueous extract which may enhance the sperm maturation in rat. A similar research also reported the increase in testicular protein after the administration of Mondia. whitei roots extract to male rats (Watcho et al., 2004; Mbongue et al., 2010).
Cholesterol is the major substrate for the synthesis of testosterone, androgenic and anabolic effects of a pharmacological substance relies on cholesterol level (Carreau, 1996). Also, a normal cholesterol level is require for normal testicular function. The increase in testicular cholesterol concentration by the extract may be attributed to the enhanced steroidogenesis. Such stimulation of steroidogenic pathway will lead to increased androgen concentration in male rats (Bedwall et al., 1994; Koloko et al., 2019). The elevated levels of testicular cholesterol observed in this study for the aqueous extract is also an indication of androgenic and anabolic affect as reported into a previous finding by Kamtchouing et al, (2002). In addition, we also observed in this study the increased activity of widely distributed acid phosphatase enzyme in the prostate and testes which is important in sperm physiology. This increase acid phosphatase activity by the extract confirmed the androgenic effects of this extract in rats (Breton et al., 1996).
Previous research has revealed that the nitrites can be considered as a reserve form of NO in blood and tissues (Okamoto et al., 2005; Jońca et al., 2015). This study investigated the level of penile nitrite as a maker of corporal NO production which has recently emerged as the most likely neurotransmitter in intracavernosal smooth muscle relaxation during penile erection (Nozaki et al., 1993). Its production and activity in the cavernosal vasculature is androgen-dependent. Aqueous extract of A. florbunda at the dose of 150 mg/kg have increased the level of penile nitrites that is, it has increased the expression of corporal NO. This enhanced corporal NO production confirmed the presence of androgenic bioactive components in aqueous extract of A. floribunda stem bark also justified the previous results obtained showing the aphrodisiac properties this extract (Kada et al., 2012; Kada et al ., 2013; Brusotti et al., 2016).
Leydig cells under luteinizing hormone (LH) stimulation produced testosterone and both are hormonal markers of androgenicity (Walton et al., 1995). The major action of testosterone is to facilitate sperm maturation during the process of spermiogenesis and to promote growth, maintenance and secretory activities of reproductive organs in male (O’Donnell et al., 1994). The increase in testosterone concentration is due to the stimulation in the synthesis of the hormone by the Leydig cells, since these cells are the main source of testosterone in rats (De Krester, 1987). It is implies that the bioactive components of the extract stimulated the mechanism intervening in the process of the hormone synthesis in the Leydig cells (Moundipa et al., 1999, Koloko et al., 2019) or through the inhibition of the aromatase (Kline and Karpinski, 2008). Also, the observed elevated serum and testicular testosterone in this finding after the treatment with the extract can be the result of the increase in cholesterol concentration which is the starting material for androgen biosynthesis. A minimum level of blood androgen is required for the maintenance of body shape, muscle mass and strength, normal sexual desire, nocturnal penile tumescence, and non-erotic penile erections in most men. Therefore, the increase in testosterone by the extract is an indication of androgenic potential of the plant. It may be responsible for the enhanced male sexual behavior observed with this extract previously (kada et al, 2012, 2013).
It is well known that the secretion of gonadotropins (LH and FSH) is controlled by hypothalamic (GnRH) and testicular hormones (Gharib et al., 1990). FSH is necessary for the initiation and maintenance of spermatogenesis while luteinizing hormone stimulates the synthesis and secretion of testosterone. The increase in the testosterone level could be due to the stimulation of the GnRH-LH signaling pathway as the extract also induced the increase of the LH level. Thus, high levels of gonadotropins and serum testosterone can be explained by a reduction or the blockage in the negative feedback of steroid hormones on pituitary gonadotropins by the extract (Malini et al., 1999). The increase in LH level caused by the aqueous extract of A. floribunda stem bark would have stimulated the production of testosterone as observed in this study. The pro-gonadotropic effect of A. floribunda aqueous extract is an indication of the ability of this extract to wisthand in the normal functioning of Sertoli cells, which in turn could stimulate the maturation of spermatozoa. The increased concentration of hormones is an indicator of a direct action on testicular hormonogenesis. Sufficient gonadotropin levels and increased testicular activity are signs of androgenicity. Thus, both androgenic and anabolic properties of the aqueous extract of A. floribunda have been proven in rats.
Androgens are increasing the number of red blood cells and the level of hemoglobin in animals (Bachman et al., 2014)). The same parameter increased for the lower concentration (150 mg/kg) of the extract dose after the treatment of the animals. This result supports the hypothesis according to which this extract would have androgenic effects.
The androgenic and anabolic effects of the aqueous extract of A. floribunda as well as its aphrodisiac properties, which is important at the dose of 150 mg/kg, could suggest a saturation of the receptors of the bioactive compounds present in the plant at the high dose (300 mg/Kg). This may be due to the competition between the bioactive phytochemical compounds hydroxyl groups’ hydrogen bond formation and the cells or organs.
Histologically, the testicular and epididymal sections of the animals treated with the extract for eight days showed the normal spermatogenesis with numerous spermatozoa in the lumen of the seminiferous tubules when compared to the control. These results suggested that the active principle of A. floribunda has no adverse effect on the testicular and epididymal morphology of animals. The significant increase in the thickness and diameter of the seminiferous tubules of animals treated with aqueous extract at a dose of 150 mg/kg compared to animals receiving distilled water would indicate maturation of the seminiferous tubules, an increase of the volume of the testes and consequently an increase in spermatogenesis and steroidogenesis. These changes confirmed once again the androgenic activity of A. floribunda aqueous extract.
Several studies have shown that saponis and flavonoids contribute to the increase of endogenous testosterone by stimulation of LH production (Hodeck et al., 2002; Temraz et al., 2006). Also, flavonoids may increase testosterone level by inhibiting Cytochrome P450 of enzyme aromatase responsible for the conversion of testosterone to oestrogen (Hodeck et al., 2002). Most of these compounds are having the hydroxyl functional groups which are acting as pharmacophore when administered as an aqueous extract in animals. Highly soluble compounds may be responsible for the androgenic and anabolic effect in rats. When they are administered in combined form at low concentrations, they showed the significant results. But, the higher concentrations may not be adsorbed with the cells or organs. Also, High Phase Liquid Chromatography couple to Electrospray Ionisation and Mass Spectrometry (HPLC-ESI-MS) apply to A. floribunda extract led to the isolation and charaterization of 4 biflavonoides known as Fukugiside, Morelloflavone, spicataside and Volkensiflavone (Brusotti et al., 2016) which may be responsible of the androgenic and anabolic effect of the extract.
Conclusion
The in-vivo results of the investigation of the androgenic effects of the aqueous extract of A. floribunda stem bark have revealed that the 150 mg/kg dose seems to be the therapeutic dose when compared with the 300 mg/kg dose. Compilation of these results suggested that this plant extract would have androgenic activity by stimulating the secretions of almost all androgen-dependent organs as well as by increasing the weight of these organs after eight days of treatment. The results of this study support the traditional use of this plant as an aphrodisiac or to address male fertility issues.
Acknowledgments
The authors gratefully acknowledged the laboratory “le bon diagnostic” of Douala, Cameroon for the hormonal assay and the laboratory of Animal Physiology of the University of Yaoundé 1 for their technical assistance.
Conflict of interest
Authors wish to confirm that there are no known conflicts of interest associated with this publication and there has been no significant financial support for this work that could have influenced its outcome.
References
Ackah AF, Tabiri HI, Alake J, Ahlidja W, Amoani B, Ofori GE, Asante-Kyei B, Temitayo GI, Adokoh CK. 2021. Allanblackia floribunda Seed extract Attenuates the ethanol-Induced gastric ulcer in Rats via the Inhibition of TNF-α and INF-γ Levels and Modulation in the expression of Ki67 Protein. Article ID 6694572. https://doi.org/10.1155/2021/6694572.
Bachman E, Travison TG, Basaria S, Davda MN, Guo W, Li M, Westfall JC, Bae H, Gordeuk V, Bhasin S. 2014. Testosterone induces erythrocytosis via increased erythropoietin and suppressed hepcidin: Evidence for a New Erythropoietin/Hemoglobin Set Point. The journals of gerontology. Series A, Biological sciences and medical sciences, 69(6):725–735.
Baskin LS, Sutherland RS, Di Sandro MJ, Hayward SW, Lipschutz J, Cunha GR. 1997. The effect of testosterone on androgen receptors and human penile growth. The Journal of Urology, 158:1113.
Bedwall RS, Edwards MS, Katoch M, Bahuguna A, Dewan R. 1994. Histological and biochemical changes in testes of zinc deficient BALB/C strain of mice. Indian Journal of Experimental Biology, 32:243-247.
Bradford MM. 1976. A rapid and sensitive method for the quantitation microgram quantities of proteins utilizing the principle of protein binding. Analytical Biochemistry, 72:248-254.
Brusotti G, Papetti A, Serra M, Temporini C, Marini E, Orlandini S, Kada SA, Watcho P, Kamtcouing P. 2016. Allanblackia floribunda Oliv: An aphrodisiac plant with vasorelaxant properties. Journal of Ethnopharmacology, 192(4):480-485.
Burris AS, Banks SM, Carter CS, Davidson JM, Sherins RJ. 1992. A long-term, prospective study of the physiologic and behavioral effects of hormone replacement in untreated hypogonadal men. Journal of Andrology, 13:297-304.
Carani C, Granata AR, Fustini MF, Marrama P. 1996. Prolactin and testosterone: their role in male sexual function. International Journal of Andrology, 19(1):48-54.
Carreau S. 1996. Paracrine control of human Leydig cell and Sertoli cell functions. Folia Histochema et Cytobiologica, 34(3-4):111-119.
De Krester DM. The testis, in: Austin, CR, Short RV, eds, A Hormonal Control of Reproduction. New York, Academic Press 1987 pp. 76–90.
Gharib SD, Wierman ME, Shupnik MA, Chin WW. 1990. Molecular biology of the pituitary gonadotropins. Endocrine Reviews, 11(1):177-199.
Gornal AG, Bardwil GS, David MM. 1949. Determination of serum proteins by the means of biuret reactions. Journal of Biological Chemistry, 177:751-766.
Gupta RS, Kachhawa JB, Chaudhary R. 2004. Antifertility effects of methanolic pod extract of Albizzia lebbeck. Asian Journal of Andrology, 6(2):155-159.
Hodeck P, Trefi P, Stibozova M. 2002. Flavonoids-potent and versatile biologically active compounds interacting with cytochromes P450. Chemico-Biological Interactions, 139(1):1-21.
Ikeda U, Takahashi M, Shimada K. 2003. C-reactive protein directly inhibits nitric oxide production by cytokine-stimulated vascular smooth muscle cells. Journal of Cardiovascular Pharmacology, 42(5):607-611.
Jońca J, Żuk M, Wasąg B, Janaszak-Jasiecka A, Lewandowski K, Wielgomas B, Waleron K, Jasieck J. 2015. New Insights into Butyrylcholinesterase Activity Assay: Serum Dilution Factor as a Crucial Parameter. PLoS ONE, 10: e0139480. https://doi.org/10.1371/journal. pone.0139480.
Kada SA, Miegueu P, Bilanda DC, Faleu NNM, Watcho P, Dzeufiet DPD, Kamtchouing P. 2013. Ejaculatory activities of Allanblackia floribunda stems bark in spinal male rats. Pharmaceutical Biology, 51(8):1014-1020.
Kada SA, Mieugeu P, Dzeufiet DPD, Faleu NNM, Watcho P, Dimo T, Kamtchouing P. 2012. Effect of aqueous extract of Allanblackia floribunda (Oliver) stem bark on sexual behaviour in adult male rats. World Journal of Pharmacy and Pharmaceutical Sciences, 1(2):585–600.
Kamtchouing P, Mbongue-Fandio GY, Dimo T, Jatsa HB. 2002. Evaluation of androgenic activity of Zingiber offcinale and Pentadiplandra brazzeana in male rats. Asian Journal of Andrology, 4(4):299-301.
Kasturi M, Manivannan B, Ahmed NR, Shaikh PD, Pattan KM. 1995. Changes in epididymal structure and function of albino rat treated with Azardirachta indica leaves. Indian Journal of Experimental Biology, 33(10):725-729.
King SR, Manna PR, Ishii T, Syapin PJ, Ginsberg SD, Wilson K, Walsh LP, Parker KL, Stocco DM, Smith RG, Lamb DJ. 2002. An essential component in steroid synthesis, the steroidogenic acute regulatory protein, is expressed in discrete regions of the brain. Journal of Neuroscience, 22 (24):10613-10620.
Kline LW, Karpinski E. 2008. Testosterone and dihydrotestosterone inhibit gallbladder motility through multiple signaling pathways. Steroids, 73 5(11):1174-1180.
Koloko BL, Bushra I, Wankeu‐Nya M, Ngaha NMI, Kenmogne H, Nzeubang NDC, Nzangueu CB, Dimo T, Dongmo AB, Lembe DM. 2019. In vivo effects of Rauvolfia vomitoria (Apocynaceae) ethanolic extract on sexual performance and reproductive activity in male rats. Andrologia, 52(1):1-12.
Lin MF, Clinton GM. 1986. Human prostatic acid phosphatase has phosphotyrosyl protein phosphatase activity. Biochemical Journal, 235(2):351–357.
Malini T, Manimaran RR, Arunakaran J, Aruldhas MM, Govindarajulu P. 1999. Effect of piperine on testis of albino rats. Journal of Ethnopharmacology, 64(3):219-225.
Mbongue GYF, Kamtchouing P, Dimo T. 2010. Effects of the aqueous extract of dry seeds of Aframomum melegueta on some parameters of the reproductive function of mature male rats. Andrologia, 44(1):53-58.
Means AR. 1975. Biochemical effects of follicle stimulating hormones on the testis, in: Greep RO, Ashwood EB, eds, Handbookof Physiology and Endocrinology. Washington DC, American Physiological Society, pp. 203-219.
Meisel RL, Sachs BD. 2001. The physiology of male sexual behavior. In: Knobil E, Neil JD, eds. The physiology of reproduction. New York: Raven Press, PP:3-105.
Mills TM, Lewis RW. 1999. The Role of Androgens in the Erectile Response: A 1999 Perspective. Molecular Urology; 3(2):75-86.
Mills TM, Reilly CM, Lewis RW. 1996. Androgens and penile erection: A review. Journal of Andrology, 17(6):633-638.
Moundipa FP, Kamtchouing P, Koueta N, Tantchou J, Foyang NPR, Mbiapo FT. 1999. Effect of aqueous extract of Hibiscus macranthus and Basella alba in mature rat testes function. Journal of Ethnopharmacology, 65:133-139.
Nozaki K, Moskowitz MA, Maynard KI, Koketsu N, Bredt DS, Snyder SH. 1993. Possible origins and distribution of immunoreactive nitric oxide synthase-containing nerve fibers in cerebral arteries. Journal of Cerebral Blood Flow and Metabolism, 13(1):70-79.
O’Donnell L, McLachlan RI, Wreford NG, Robertson DM. 1994. Testosterone promotes the conversion of round spermatids between stages VII and VIII of the rat spermatogenic cycle. Endocrinology, 135(6):2608-2614.
Okamoto M, Tsuchiya K, Kanematsu Y, Izawa Y, Yoshizumi M, Kagawa S, Tamaki T. 2005. Nitrite-Derived Nitric Oxide Formation Following Ischemia-Reperfusion Injury in Kidney. American Journal of Renal Physiology, 288 (1):182-187.
OMS. 1993. Manuel de laboratoire de l’OMS: Analyse du sperme humain et de l’interaction des spermatozoïdes avec le mucus cervical. Les éditions INSERM, Paris, PP:85-89.
Sandroni P. 2001. Aphrodisiacs past and present: A historical review. Clinical Autonomic Research, 11(5):303-307.
Shabsigh R. 1997. The effects of testosterone on the cavernous tissue and erection. World Journal of Urology, 15(1):21-26.
Steidle C, Schwartz S, Jacoby K, Sebree T, Smith T, Bachand R. 2003. AA2500 testosterone gel normalizes androgen levels in aging males with improvements in body composition and sexual function. Journal of Clinical Endocrinology and Metabolism, 88(6):2673- 2681.
Temraz A, El Gindi OD, Kady HA, De Tommasi N, Braca A. 2006. Steroidal saponins from the aerial parts of Tribulus alatus Del. Phytochemistry; 67(10):1011-1018.
Tetsatsi, ACM, Nkeng-Effouet PA, Alumeti DM, Bonsu FGR, Kamanyi A, Watcho P. 2019. Colibri® insecticide induces male reproductive toxicity: alleviating effects of Lannea acida (Anacardiaceae) in rats. Basic and Clinical Andrology, 29:16.
Walton S, Cunliffe WJ, Kaczkes K, Early AS, McGarrigle HHG, Katz M, Reese RA. 1995. Clinical ultrasound and hormonal markers of androgenicity in acne vulgaris. British Journal of Dermatology, 133 (2):249-253.
Watcho P, Kamtchouing P, Sokeng SD, Moundipa PF, Tantchou J, Essame JL, N Koueta. 2004. Androgenic effect of Mondia whitei roots in male rats. Asian Journal of Andrology, 6(3):269-272.
Yakubu MT, Oyeyipo TO, Quadri AL, Akanji MA. 2013. Effects of aqueous extract of Musa paradisiaca root on testicular function parameters of male rats. Journal of Basic and Clinical Phisiology and Pharmacology, 24(2):151-157.
Yildrim MK, Yildrim S, Utkan T, Sarioglu Y, Yalman Y. 1997. Effects of castration on adrenergic, cholinergic and nonadrenergic noncholinergic responses of isolated corpus cavernosum from rabbit. British Journal of Urololgy, 79(6):964-970.