

Eric Martial Deutchoua Ngounou*, Faustin Dongmo, Yannick Dimitry Mang, Slestin Sokeng Dongmo, Nicolas Njintang Yanou
Department of Biological Sciences, Faculty of Science, University of Ngaoundere, Cameroon
*Address for Corresponding Author
Eric Martial Deutchoua Ngounou
Department of Biological Sciences, Faculty of Science, University of Ngaoundere, Cameroon
Abstract
Objective: Clerodendrum thomsoniae is an important underutilized plant with established lipid-lowering, anti-free radicle, hypoglycemic and antidiabétic effect. The present study was aimed at evaluating the acute and subchronic toxicity in experimental animals. Materials and methods: Experiments were conducted according to OCDE425 and OCDE407 guidelines for the study of acute and subchronic toxicity. Acute toxicity studies used female and male mices Mus musculus by single dose oral administration of aqueous extract of C. thomsoniae (AECT) at doses of 2000, 5000 and 9000 mg/kg. Subchronic toxicity study was carried out during 28 days by daily oral administration of single dose of AECT on albino rats (male and female). The control group received vehicle (10 mL/kg b.m.) while treated groups (II, III and IV) were administrated AECT at the respective doses of 312.5, 625 and 1250 mg/kg. Results: A single dose oral administration of aqueous extract of C. thomsoniae till 9000 mg/kg did not lead to death and changing in the behavior 14 days observation after administration. However, slight change was observed concerning motility 3hours after oral administration of extract dose more than 6000 mg/kg. Daily administration for 28 days of the extract at the therapeutic dose of 1250 mg/kg did not bring any significant changes on the relative mass of organs, transaminases activities and creatinine level. Conclusion: The study showed that with an LD50 greater than 9000 mg/kg and a potential hepatoprotective effect, the aqueous extract of C. thomsoniae leaves is favorable to the production of an improved traditional medicine, after preclinical and clinical tests.
Keywords: Clerodendrum thomsoniae Linn, leaves, acute toxicity, subchronic toxicity
Introduction
Paracelsus (1493-1541) reported that all substances are poisons, only the right dose can distinguish a poison from a drug. Toxicity is the intrinsic ability of a chemical or physical agent to have a harmful effect on an organism. It is the capacity specific to various substances whose absorption of single or repeated doses has the effect of disturbing the metabolism of living beings, causing physiological disorders which can go as far as the death of exposed individuals (Ramade, 2002). According to Stéphane Malard (2018), the harmful effect is any alteration due to treatment deviating from normality taken as a reference, which reduces the ability of an organism to survive, reproduce or adapt to the environment.
The toxicity of a substance in the body depends on the nature of the substance, the dose and the duration of exposure, the various factors linked to the individual (sex, age, nutritional and hormonal status), environmental factors and simultaneous or prior exposure to other chemicals.
Individual factors can modify the absorption, excretion, metabolic transformations and sensitivity of the receptor in the target organ (Bayiha, 2011). The study of the toxicity of a substance is the set of pharmacological tests, which determine the degree or harmfulness of the latter in order to regulate its use. The action of a toxic substance is evaluated according to several parameters, including its mode of administration (oral, intravenous, intraperitoneal), the dose administered, the observed mortality rate, weight change, histology of certain organs, the modification of certain biochemical parameters of the blood called toxicity markers such as transaminases (ALAT, ASAT), bilirubin, creatinine, urea (Serrano, 1990). Ethnomedical importance of various species of Clerodendrum genus has been reported in various indigenous systems of medicines and folk medicines.
Indeed, studies highlight the short, medium- and long-term toxic effects of plant extracts with established biological properties (Ayoola et al., 2008). Plants have been used for centuries as remedies for human diseases because they contain components of therapeutic value (Arthur et al., 2011). The use of plants in saving human beings has a long history across different cultures in the world (Hosseinzadeh et al., 2015; Silva et al 2019) At present, natural substances in plants are still the primary reservoir of new drugs. They represent almost 60% of the drugs we have. The remaining 40% or synthetic drugs are often born from the chemical synthesis of molecules or parts of natural molecules taken as the head of the series (Fouché et al., 2000). Today, the practice of herbal medicine in conventional medicine has drawn more attention and is becoming accepted globally (Silva et al 2019). Present estimates indicate that about eighty percent of the world’s population relies on traditional medicine for health care delivery (Akah, 2008). It is in this sense that we are interested in Clerodendrum thomsoniae which is a species much better known for its ornamental aspect and cultivated for that in India (Kar et al., 2014). In Ngaoundere, Cameroon, the leaves of C. thomsoniae are used in the treatment of intestinal worms, hypertension and diabetes.
The antioxidant and lipid-lowering properties of this plant have been established (Deutchoua et al., 2020). Phytochemical analyzes have revealed the presence of flavonoids, glucosides, phenolic compounds, lipids, steroids and alkaloids (Deutchoua et al., 2020; Kar et al., 2019). The genus Clerodendrum has more than 400 species of plants of all types: trees, shrubs, herbs, climbers. Most of them live in tropical regions with a hot climate. However, no studies on the toxicity of C. thomsoniae leaves have been described in the literature. Therefore, in the present investigation, we aimed to assess the toxicity (acute and subchronic) in mice and rats, respectively, to evaluate the safety of the oral administration of the aqueous extract of the leaves of C. thomsoniae (AECT) in order to increase the confidence in their safety to humans to treat various ailments.
Materials and methods
Sampling and production of Clerodendrum thomsoniae powder
Clerodendrum thomsoniae leaves used for this study were collected from Mbideng, a neighborhood of Ngaoundéré, in the Adamawa Region of Cameroon. The plant was identified and authenticated at the national herbarium, (NO Letouzey 11090 from the herbarium collection NO 28476 / SRF / Yaounde-Cameroon), a voucher specimen (N° 28476 / SRF / Yaounde-Cameroon) was retained in laboratory of biology (Faculty of Science; Ngaoundere University) for further reference. Young mature leaves were carefully cleaned, sorted, and dried in a ventilated electric rotary dryer (Riviera & Bar brand) at 40 ± 2 ° C for 48 hours. After drying, the leaves were crushed to make a fine powder using an electric grinder (Culatti, Polymix, France) equipped with a sieve of 500 µm.
Preparation of Clerodendrum thomsoniae aqueous extract
Different quantities of powder (1.25, 2.5 and 5g) were respectively mixed with 40mL of distilled water. The different mixtures were placed in a water bath at 70 ± 2°C and extracted for 30min while stirring then, cooled for 30min and centrifuged at 1500 rpm/15 min at 20°C using a refrigerated centrifuge. The supernatant was collected and the residue was dissolved in 40 mL and re-extracted as mentioned above. The supernatants were combined and concentrated under vacuum in a rotary evaporator and dried in a desiccator at 40 °C. The crude extract was weighed and used to prepare 31.25, 62.5 and 125mg/mL of aqueous extract of Clerodendrum thomsoniae (AECT) corresponding to the 3 doses administrated (312.5, 625 and 1250mg/kg).
Experimental animals
Healthy male albino rats aged of 6 to 7 weeks weighing between 150 to 200g as well as the Swiss Mus musculus strain mice aged 10 to 12 weeks weighing between 26 and 30g, were raised in the animal house of the Faculty of Science of the University of Ngaoundéré (Cameroon) at an ambient temperature of 22 ± 3°C and a relative humidity of 54 ± 2% under a 12h/12light/dark cycle. During acclimatization to laboratory conditions for four days, animals were randomized into experimental and control groups and housed individually in sanitized polypropylene cages housed with sterile paddy husk as bedding. Animals were given free access to standard pellet diet and water ad libitum. Experiment was conducted according to the directives of the Committee for the control and supervision of animal experiments (CPCSEA) and ethically approved by the Institutional Committee of the Ministry of Scientific Research and Innovation of Cameroon.
Acute oral toxicity study
The study was carried out in accordance with the OECD guideline 425 "dose adjustment" method (OECD, 2008a) with some modifications. The objective was the evaluation of the lethal dose (LD50), and the results make it possible to classify a substance for acute toxicity according to the Globally Harmonized System of Classification and Labeling of Chemicals. It consisted in testing three doses of AECT (2000 mg/kg, 5000 mg/kg and 9000 mg/kg) on 24 mice (12 males and 12 females) Mus musculus after fasting period of 15 hours. Male and female mice were divided into 04 groups of 06 mice each with females separated from the males: a normal control group consisting of 3 females and 3 males receiving distilled water at the dose of 10 mL/kg and each of the three doses of AECT were given to the three other groups. Animals were daily fed during the period of treatment. The animals were observed for signs of toxicity and mortality for the first critical 3 hours and thereafter daily for 2 weeks. The behavior and the number of deaths over a period of 14 days were evaluated. Signs of toxicity included modification of the coat, motility, tremors, mass, grooming, breathing, sensitivity to noise after a metallic shock, the appearance of stools, mobility as well as death were observed. The oral median lethal dose (LD50) was calculated as the geometric mean of dose that caused 0 % and 100 % mortality respectively.
Subchronic toxicity study
Subchronic oral toxicity study was performed according to the Organization of Economic Co-Operation and Development (OECD) guideline 407 for testing of chemicals (OCDE, 2008b).
Based on the results of acute toxicity study that indicated AECT was nontoxic at the dose level of 9000 mg/kg, 24 wistar rats (12 males and 12 females) were divided into four groups of 6 animals each (males isolated from females) with 3 males and 3 females as follows: group 1 (normal control), receiving distilled water 10 mL/1kg of body mass; groups 2, 3 and 4 receiving an AECT respectively at doses of 312.5; 625; 1250 mg/kg body mass. During the treatment of 28 days, various toxic signs and observations, such as body mass, mortality, food and water intakes were monitored. Rats were fed and had access to water ad libitum. The percent change in body mass was calculated using the formula:
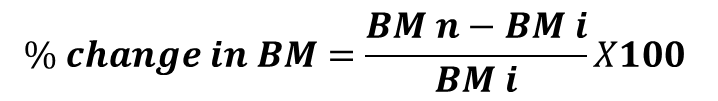
BM = Body Mass
BMn = Body Mass at the end of treatment
BMi = Body Mass on the first day
At the end of the treatment, the rats were fasted overnight and anesthetized (with ketamine, at dose of 50 mg/kg) for 24 h, The serum from non-heparinized blood samples were collected for determining biochemical analysis. Rats were then euthanized after blood collection and the internal organs (heart, liver, kidney, and lungs) were removed, rinsed with 0.9% salt solution and weighed to determine the relative organ mass. Relative mass of each was calculated according to the formula:
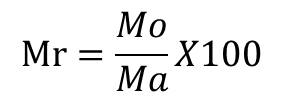
Mr= relative Mass of organ (g/100 g)
Mo= Absolute mass of organ removed (g)
Ma= Body Mass of animal at sacrifice (g)
Biochemical analysis
Blood collected in non-heparinized tubes were then centrifuged at 3000 rpm for 10 min. Several biochemical parameters were analysed in the serum separated, in particular creatinine by the colorimetric method of Jaffé (Etame et al., 2017) using the CREATININE LR SGMitalia kit, alanine aminotransferase by the kinetic method UV IFCC optimized (Etame et al., 2017) using the GPT-ALT LR GSMitalia kit and aspartate aminotransferase by the kinetic method UV IFCC optimized, using the GOT-AST LR GSMitalia kit The first absorbance of the samples was read against the blank at 340 nm and the second absorbance was read 30 s later, at the same wavelength and the urea concentration determined by the formula:[Creatinin](mg/dl) = ∆DO Sample/ ∆DO Standard × [Standard] (mg/dl); The enzymatic activity of aspartate aminotransferase was obtained from the following formula: Activity (U/L) = ∆A/mn x 1746 ∆A = Absorbance variation between 2-time intervals; ∆A/mn = variation of the absorbance of the sample per minute; 1746 = multiplication coefficient. The enzymatic activity of alanine aminotransferase was obtained according to the formula: Activity (U/L) = ∆A/mn x 1746; ∆A = Absorbance variation between 2-time intervals; ∆A/mn = variation of the absorbance of the sample per minute; 1746 = multiplication coefficient. Total cholesterol was assessed according to Roeschlau et al. (1974) using the Randox Laboratories UK commercial kit.
Statistical analysis
The results are expressed as mean ± SD (n=4). The data was evaluated by one-way analysis of variance (ANOVA) using SPSS version 20 and Excel 2016. P values < 0.05 were considered statistically significant.
Results
Acute oral toxicity study
In oral acute toxicity studies, no untoward clinical signs were observed in the rats at all the doses studied (100, 1000, 2500 and 5000 mg). There were no change in the nature the coat, motility, tremors, mass, grooming, breathing, sensitivity to noise after a metallic shock, the appearance of stools as shown in table 1. No mortality was observed at all doses from the critical 48 hours post administration to the end of the fourteen day (Table 1). Orally, 9000 mg/kg of AECT was well tolerated in mice even after 14 days. Meanwhile, At the dose 9000 mg / kg, a decrease in locomotion has been observed in animals, which is characterized by agglutination of the animals in one place which can make one think of signs of toxicity. The stool has become mushy and exploration has decreased. These results showed that in single dose, there are no adverse effects of AECT, indicating that the medium lethal dose (LD50) is higher than 9000 mg/kg for both male and female mice (orally).
Table 1. Effects of the aqueous extract of C. thomsoniae on some physical and physiological parameters in mice according to the doses after 3 h, 48 h and 14 days
|
Parameters |
Dose (mg / kg) |
|||||
|
0 |
2000 |
|
5000 |
9000 |
||
|
Behavior 3h later |
Grooming |
N |
N |
|
N |
N |
|
Motility |
N |
N |
|
N |
D |
|
|
Tremor |
N |
N |
|
N |
N |
|
|
Noise sensitivities |
N |
N |
|
N |
N |
|
|
Coat |
N |
N |
|
N |
N |
|
|
Stool appearance |
N |
N |
|
N |
N |
|
|
Deaths |
0 |
0 |
|
0 |
0 |
|
|
Behavior 48h later |
Grooming |
N |
N |
|
N |
N |
|
Motility |
N |
N |
|
N |
N |
|
|
Tremor |
N |
N |
|
N |
N |
|
|
Noise sensitivities |
N |
N |
|
N |
N |
|
|
Coat |
N |
N |
|
N |
N |
|
|
Stool appearance |
N |
N |
|
N |
P |
|
|
Deaths |
0 |
0 |
|
0 |
0 |
|
|
Behavior after 14 days |
Grooming |
N |
N |
|
N |
N |
|
Motility |
N |
N |
|
N |
N |
|
|
Tremor |
N |
N |
|
N |
N |
|
|
Noise sensitivities |
N |
N |
|
N |
N |
|
|
Coat |
N |
N |
|
N |
N |
|
|
Stool appearance |
N |
N |
|
N |
N |
|
|
Deaths |
0 |
0 |
|
0 |
0 |
|
N : Normal ; D : Decrease ; P : Pasty
Subchronic toxicity test
Food intake
During the 28 days of oral administration of AECT, rats were fed a standard diet. Figure 1 the effect of AECT on food intake. It emerges from this figure that in general, the food intake increased during the first 3 weeks of treatment with AECT with greater increase in the animal control group receiving the distilled water. During the fourth week of treatment with AECT, a decrease in food intake was observed whatever the group. However, food intake remained higher than that of the first week. Generally, in the control group, the increase in food intake is 18.82% compared to 19.37%, 15.87% and 20.32% for the treated groups with the different doses of AECT respectively 312.5 mg / kg, 625 mg / kg and 1250 mg / kg. Despite these increases, no significant difference was observed between the groups.
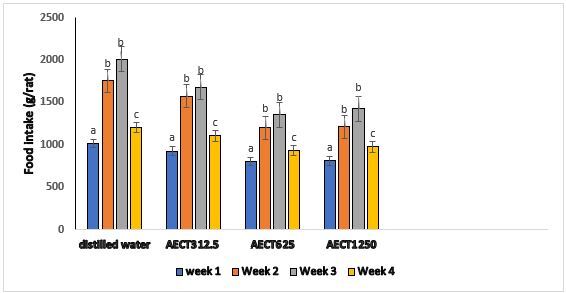
Figure 1. Effect of the AECT on the food intake during treatment
Weekly body weight
The effect of the AECT on the body mass of the animals (males and females) compared with vehicle is as shown in Figure 2 and 3. There were significant increases in the body mass of males treated with AECT. However, there was no significant increase in percent body mass change in female rats treated with AECT at doses of 312.5 mg / kg and 625 mg / kg on day 28 (p<0.05). For female rats, significant increases were observed at 1250 mg / kg on day 28 (p<0.05).
Relative organ weights after 28 days of treatment by AECT are shown in Tables 2 and 3. There was no significant difference compared to control in relative organ weights in both sexes.
Table 2. Effect of the AECT on the relative organ weights of male rats
|
Groups |
Liver |
Kidney |
Lungs |
Heart |
|
Distilled water |
2,89±0,81a |
0,78±0,13a |
0,95±0,14a |
0,44±0,18a |
|
AECT312,5 |
2,79±0,32a |
0,71±0,61a |
0,89±0,42a |
0,41±0,13b |
|
AECT625 |
2,88±0,26a |
0,75±0,14a |
0,90±0,21a |
0,42±0,23a |
|
AECT1250 |
2,91±0,22a |
0,79±0,15a |
0,96±0,17a |
0,43±0,14a |
Values were expressed as mean ± standard deviation; n = 3. On the same column, the means with different letters are significantly different (P <0.05). AECT = Aqueous extract of Clerodendrum thomsoniae at different doses (312.5; 625 and 1250 mg / kg)
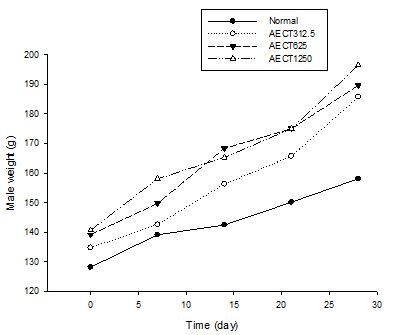
Figure 2. Effect of the AECT on the body weight of male during the treatment
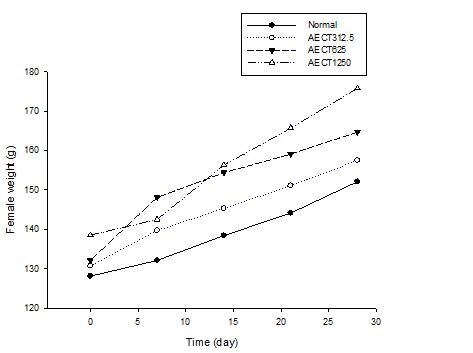
Figure 3. Effect of the AECT on the body weight of female during the treatment
Table 3. Effect of the AECT on the relative organ weights of female rats
|
Groups |
Liver |
Kidney |
Lungs |
Heart |
|
Distilled water |
2,86±0,61a |
0,74±0,17a |
0,85±0,18a |
0,42±0,21a |
|
AECT312,5 |
2,63±0,39a |
0,77±0,12a |
0,82±0,31a |
0,44±0,14a |
|
AECT625 |
2,87±0,28a |
0,78±0,15a |
0,86±0,15a |
0,41±0,33a |
|
AECT1250 |
2,94±0,23a |
0,75±0,14a |
0,87±0,02a |
0,44±0,12a |
Values were expressed as mean ± standard deviation; n = 3. On the same column, the means with different letters are significantly different (P <0.05). AECT = Aqueous extract of Clerodendrum thomsoniae at different doses (312.5; 625 and 1250 mg / kg).
Biochemical analysis
Figure 4 explores the total cholesterol level of normal and AECT treated animals after the 28 days experimental period. The results revealed that the extract does not adversely alter the total cholesterol level of the animals after subchronic treatment. No significant difference in serum total cholesterol levels in rats treated with AECT for all the dose compared to control
The unaltered values of renal biochemical parameters (creatinine), as shown in Table 4, indicate that subchronic treatment with AECT did not possess significant adverse effect on renal functioning of the animals. No significant difference in the liver function parameters like alanine aminotranferase (ALT), aspartate aminotransferase (AST) was observed between these various treatments. We observed a slight decrease which remained non-significant in ALAT and ALSAT levels for the both male and female.
Table 4a. Effect of AECT on biochemical profiles of the control and treated animals in the subchronic toxicity study in female rats
|
Groups |
ALAT(UI/L) |
ASAT(UI/L) |
CREATININE (mg/L) |
|
Normal |
105,3±25,4a |
249,6±81,9b |
8,9±0,7c |
|
AECT312,5 |
92,6±30,6b |
222,9±64,4a |
8,5±0,7c |
|
AECT625 |
94,3±21,4b |
228,3±78,3c |
8,8±0,7c |
|
AECT1250 |
96,7±23,3b |
236,7±83,6d |
8,9±1,0c |
Table 4b. Effect of AECT on biochemical profiles of the control and treated animals in the subchronic toxicity study in male rats
|
Groups |
ALAT(UI/L) |
ASAT(UI/L) |
CREATININE (mg/L) |
|
Témoin |
108,5±22,1a |
252,6±79,7b |
9,7±0,7c |
|
EACT312,5 |
95,7±28,4b |
223,5±63,1a |
8,8±0,7c |
|
EACT625 |
98,5±24,2b |
232,1±68,5c |
9,3±0,7c |
|
EACT1250 |
97,1±21,2b |
240,4±76,6d |
9,1±1,0c |
Values were expressed as mean ± standard deviation; n = 3. On the same column, the means with different letters are significantly different (P <0.05). AECT = Aqueous extract of Clerodendrum thomsoniae at different doses (312.5; 625 and 1250 mg / kg)
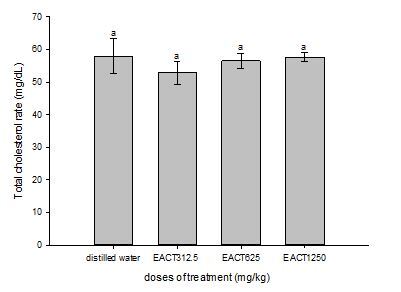
Figure 4. Effect of treatment in total cholesterol level on rats (male & female)
Discussion
In recent decades, the use of herbal medicines has experienced a phenomenal resumption not only in developing countries but also in developed countries (Verma and Singh, 2010). The World Health Organization has also recommended the use of herbal remedies for diseases for which synthetic drugs are not available. Like any drug, the therapeutic action of plants is linked to the presence of bioactive molecules, which in high doses, create harmful effects on organs, like the kidneys (Jadeja et al., 2011). Therefore, a preclinical toxicological evaluation of plant extracts is required before any use by humans. The toxicological evaluation of the aqueous extract of C. thomsoniae makes it possible to measure its capacity to cause harmful health effects by determining its possible collateral effects to ensure safety in use by population who already use it. Experimental screening method is therefore important to ascertain the safety and efficacy of these herbal drugs.
The acute toxicity study may provide initial information on the mode of toxic action of an agent, acts as the basis for classification and labelling, and helps in deciding the dose of novel compounds in animal studies. Moreover, if a high dose (e.g., 5000 mg/kg) is found to be survivable, no further acute testing will be conducted (NRC, 2006). In the acute oral toxicity study of the extract, no noticeable change in the general behavior of rats was observed. All animals survived. The lack of mortality or toxicity at oral treatment of over 9000 mg / kg body weight obtained and according to the chemical labelling and classification of acute systemic toxicity based on oral LD50 values recommended by the Organisation of Economic Co-operation and Development (OECD, Paris, France): very toxic, ≤ 5 mg/kg; toxic, > 5 ≤ 50 mg/kg; harmful, > 50 ≤ 500 mg/kg; and no label, > 500 ≤ 2000 mg/kg (Walum, 1998), the aqueous extract of Clerodendrum thomsoniae leaves is practically nontoxic at single dose and is therefore safe for oral use in the management of several diseases.
Subchronic studies provide information on dosage regimens, target organ toxicity, and identify observable adverse effect that may affect the average life span of experimental animals. Consequently, in this study, the leaves of C. thomsoniae were evaluated in rats at doses of 312.5, 625, and 1250 mg/kg for 28 days. The body weight changes serve as a sensitive indication of general health status of animals (Hilaly et al., 2004). After 28 days of treatment of the extract, all the animals exhibited a normal increment in body weight. There was no sign of toxicity during the experimental period as compared to control. It can be stated that leaves of C. thomsoniae did not interfere with the normal metabolism of animals. The significant increment in food intake is considered as being responsible for augmentation in body mass gain. It’s may be suggested that the AECT could stimulate appetite. This stimulation of appetite could be due to his phytochemical constituents (Deutchoua et al., 2020). In the same line, no significant change in the relative mass of the heart, liver, lung, spleen and kidney was observed, suggesting that administration of AECT leaves at subchronic oral doses produced no effect on the normal growth. Our results are similar to those obtained by Pieme et al., (2006) on rats treated for 26 days with the aqueous extract of Senna alata. The results of this study revealed no significant change in the relative organ weight of control and treated groups which showed that none of the organs were adversely affected, nor showed any sign of toxicity throughout the study.
In this study, we observed a non-significant decrease in total cholesterol level, proof of its protective action against cardiovascular diseases (Law et al., 2003). Same results on cholesterolemia were obtained by Mosaddegh et al., (2004) with the aqueous extract of Paliurus spina-christi and by Sharmila et al., (2007) with the extract aqueous of Trichosanthes dioica Roxb in rats. Studies of Deutchoua et al., (2020) revealed the lipid-lowering effect of AECT in hyperlipidemic rats. These results can be justified by the inhibition in the activity of lipoprotein lipase or due to the inhibition of the 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase which is the rate-limiting enzyme of cholesterol biosynthesis or reduction in the absorption of cholesterol from the intestines. Evaluation of serum biochemistry was done to identify the possible alterations in renal and hepatic functions affected by extract. Alanine aminotransferase (ALT) is released by the liver and its increase in plasma level is an indicator of loss of functional integrity of the liver cell membrane (Watkins and Seef, 2006). The liver and heart release AST and ALT, and an elevation in the levels of these enzymes are indicators of liver and heart damage (Crook, 2006). The lack of significant alterations in the levels of ALT, AST, creatinine, which are good indicators of liver and kidney functions (Olorunnisola et al., 2012) suggests that sub-chronic administration of extract neither altered hepatocytes and kidneys of rats, nor the normal metabolism of the animals. In this study the level of ALAT and ASAT showed a slight decrease compared to the normal control. These observations indicate hepatic protective role (Gomé et al., 2012) of EACT as demonstrated for other extracts Eleophorbia drupifera (Akpanabiatu et al., 2003), Artemisia afra (Mukinda and Syce, 2007), Clerodendrum serratum (Agrawal et al., 2013). Flavonoids have demonstrated hepatoprotective properties (Barbenko & Shakhova, 2003). The phytochemical study of EACT (Deutchoua et al., 2020) revealed the presence of flavonoids in our extract. The hepatoprotective properties observed could be du to the presence of flavonoids.
Conclusion
The study showed that aqueous extract of the leaves of LD50 is more than 9000 mg / kg. We may conclude that C. thomsoniae leaf aqueous extract is not toxic at all doses studied here in and did not produce any evident symptoms in the acute and subchronic oral toxicity studies. However, assessment of toxicity by the intraperitoneal route is worth exploring, the effect of the extract on pregnant rats, foetuses, and their reproductive capacity to complete the safety profile of this plant extract. More studies need to be conducted in other to establish antitumoral and protective effect on liver using carbon tetrachloride. This could help to produce medicin.
Acknowledgements
We sincerely thank Department of Biology of the Faculty of Sciences, Ngaoundere University through the Laboratory of Medicinal plants, Health and Pharmaceutical Formulation; and the National School of Agro-Industrial Sciences (ENSAI) of the University of Ngaoundere through the Laboratory of Food Biophysics and Biochemistry and Nutrition, for extending their support in facilitating instruments and chemicals for this work.
Conflict of interest
The authors have declared that there is no conflict of interest.
References
Agrawal SK, Jat RK, Chhipa RC. 2013. Pharmacological evaluation of hepatoprotective activity of Clerodendrum serratum. International Journal of Pharmacology and Toxicology, 3:67–70.
Akah PA. 2008. Indigenous Knowledge and Medical practice. In: Ethnopharmacology. Akah PA. (eds) Research Signpost, Kerela, India, pp 1-13.
Akpanabiatu MI, Ihiri AO, Eyong EU, Eng MU. 2003. Biochemical histological effect of Eleophorbia drupifera leaf extract in wistar albino rats. Pharmaceutical Biology, 41(2):96-99.
Arthur, F.K.N., Woode, E., Terlabi, E.O., and Larbie, C.2011. Evaluation of acute and subchronic toxicity of Annona muricata(Linn.) aqueous extract in animals. European Journal of Experimental Biology, 1(4): 115-124.
Ayoola G, Coker H, Adesegun S, Adepoju-Bello A, Obaweya K, Ezennia E, Atangbayila T.2008. Phytochemical screening and antioxidant activities of some selected medicinal plants used for malaria therapy in South Western Nigeria. Tropical Journal of Pharmaceutical Research, 7(3): 1019-1024.
Bayiha JC. J.2011. Etude de la toxicité systémique de l'extrait aqueux du mélange des plantes Aframomum melegueta, Mondia whitei, Piper guineense, et Zingiber officinale chez le rat. Mémoire de Master. Université de Yaoundé, pp 1-69.
Crook MA. 2006. Clinical Chemistry and Metabolic Medicine. 7th edn. Hodder Arnold, London, pp 426.
Deutchoua EM, Mang YD, Sokeng DS, Njintang YN. 2020. Hypolipidemic and antioxidant activity of aqueous extract of Clerodendrum thomsoniae Linn. (Verbenaceae) leaves in albino rats, Rattus norvegicus (Muridae). Journal of Pharmacognosy and Phytochemistry, 9(1): 595-602.
Etame-LG, Dibong SD, Yinyang J, Elimbi M, Ngoule CC, Kidik PC, Ngene JP, Tankeu SE, Okalla EC, Ngaba GP, Nda MJP, Nnanga NE. 2018. Étude de la toxicité aigüe et subaigüe de l’extrait au vin de palme des rhizomes de Curcuma longa Linn. Journal of Applied Biosciences, 132 : 13452 – 13460.
Fouché J, Marquet A, Hambuckers A. 2000. Les plantes médicinales, de la plante au médicament : Observation du monde des plantes, pp 11.
Gome M, Kouakou K, Toure A, Traore F. 2012. Étude de la toxicité aiguë et subchronique de l’extrait aqueux de Passiflora foetida Linn. (Passifloraceae) chez les rats et les souris. International Journal of Biological and Chemical Sciences, 5(5):1777.
Hilaly J, Israili H, Lyoussi B. 2004. Acute and chronic toxicological studies of Ajuga iva in experimental animals. Journal of Ethnopharmacology, 91:43–50.
Hosseinzadeh S, Jafarikukhdan A, Hosseini A, Armand R. 2015. The application of medicinal plants in traditional and modern medicine: a review of Thymus vulgaris. International Journal Clinical Medicine, 6:635-42.
Jadeja RN, Thounaojam MC, Ansarullah, Jadav SV, Patel MD, Patel DK, Salunke SP, Padate GS, Devkar RV, Ramachandran AV. 2011. Toxicological evaluation and hepatoprotective potential of Clerodendron glandulosum.Coleb leaf extract. Human and Experimental Toxicology, 30(1): 63–70.
Kar P, Chakraborty AK, Bhattacharya M, Mishra T, Sen A. 2019. Micropropagation, genetic fidelity assessment and phytochemical studies of Clerodendrum thomsoniae Balf. f. with special reference to its anti-stress properties. Research in Plant Biology 9: 9-15.
Kar P, Goyal AK, Das AP, Sen A. 2014. Antioxidant and pharmaceutical potential of Clerodendrum L.: An overview. International Journal of Green Pharmacy, 8: 210-216.
Law MR, Walb NJ, Rudnicka AR. 2003. Quantifiying effect of statin on low density lipoprotein cholesterol, ischaemic heart disease & sto>Mosaddegh M, Khoshnood J, Kamalinejad M, Alizadeh E. 2004. Study on the effect of Paliurus spina-christi on cholesterol, glyceride and HDL level in diabetic male rats fed a high cholesterol diet. Iranian Journal of Pharmaceutical Research, 3: 51-54.
Mukinda JT, Syce JA. 2007. Acute chronic toxicity of the aqueous extract of Artemisia afra in rodents. Journal of Ethnopharmacology, 112(1) :138-144.
National Research Council (NRC). 2006. Toxicity Testing for Assessing Environmental Agents; Interim Report; National Academics Press: Washington, DC, USA.
Olorunnisola OS, Bradley G, Afolayan AJ. 2012. Acute and subchronic toxicity studies of methanolic extract of Tulbaghia violacea rhizomes in Wistar rats. African Journal of Biotechnology 11: 14934–14940.
Organization for Economic Co-operation and Development (OCDE). 2008a. Pharmacopée européenne. 6eme édition, Tome1, 178-568.
Organization for Economic Co-operation and Development (OCDE).2008b. Ligne directrice de l’OCDE pour les essais de produits chimiques 407.
Pieme CA, Penlap VN, Nkegoum B, Taziebou CL, Tekwu EM, Etoa FX, Ngongang J. 2006. Evalution of acute and subacute toxicities of aqueous ethanolic extract of leaves of Senna alata (L) Roxb (Ceasalpiniaceae). African Journal of Biotechnology, 5(3): 283-289.
Ramade F. 2002. Dictionnaire encyclopédique de l’écologie et des sciences de l’environnement édition Dunod (2èmeédition), pp 1075.
Roeschlau P, Bernt E, Gruber W. 1974. Enzymatic determination of total cholesterol in serum. Zeitschrift für klinische Chemie und klinische Biochemie, 12: 226-7.
Serrano JJ. 1990. Toxico–pharmacologie expérimentale des plantes médicinales. Actes du 1er colloque européen d’ethnopharmacologie. Office de la recherche scientifique d’outre-mer (ORSTOM), pp 210-218.
Sharmila BG, Kumar G, Rajasekara PM. 2007. Cholesterol lowering activity of the aqueous fruit extract of Trichosanthes dioica Roxb (L) in normal and streptozotocin diabetic rats. Journal of Clinical and Diagnostic Research, 1(6): 561-569.
Silva JC, Azevedo PSDS, Souza FM, Aragão KKV, Sabino CKB, Carvalho GD, Morais ICP, Mendes MB, Chaves MH, Oliveira AP. 2019. Vasorelaxant activity and acute toxicity of the ethanolic extract of Zanthoxylum rhoifolium Lam leaves. Brazilian Journal of Pharmaceutical Sciences, 55: e17754.
Stéphane M. 2018. Méthodes d’évaluation de la toxicité Effets sur la reproduction. Institut national de recherche et de sécurité. DIU toxicologie médicale, pp 50.
Verma S, Singh SP. 2010. Current and future status of herbal medicines. Veterinary World. 1: 347-350.
Walum E.1998. Acute oral toxicity. Environmental Health Perspectives, 106 (2): 497-502.
Watkins PB, Seef LB. 2006. Drug induced liver injury: Summary of a single topic research committee. Hepatology, 43: 618-631.