

Ajay Singh Kushwah1*, Ghanshyam Das Gupta2
1 PhD Research Scholar, IK Gujral Punjab Technical University, Jalandhar- 144603 (Punjab) India;and Department of Pharmacology, Amar Shaheed Baba Ajit Singh Jujhar Singh Memorial College of Pharmacy, Bela-140111, Ropar (Punjab) India.
2 Indo-Soviet Friendship College of Pharmacy, Ferozepur Road, GhalKalan, Moga-142001,(Punjab) India.
*Address for Corresponding Author
Ajay Singh Kushwah
Assistant Professor
Department of Pharmacology
ASBASJS Memorial College of Pharmacy, Bela-140111, Ropar, (Punjab) India
Tele: +91-9417459195, FAX-01881-263655
Abstract
Background: Quercetin and Metformin, showing that the antihyperglycemic effect could be attributed to AMPK stimulation followed by gluconeogenesis process. Objectives: The aim of the present study was to investigate the effect of the individual and concomitant use of metformin and quercetin for treating of diabetic state associated with cardiovascular diseases. Material and methods: Adult healthy Wistar rats (either sex) body weight 220-250g, induction of diabetes by injection of STZ (60mg/kg). Diabetic rats were randomly divided into five different groups; (C) normal vehicle control; (STZ60) control, test groups (STZ60+M200) and (STZ60+Q50); concomitant test drug administered groups (STZ60+M200+ Q50). Daily body weights were measured and end of the study blood and hemodynamic parameters evaluated. After studies, the animals were sacrificed to estimate heart biochemical and histopathological studies were carried out. Results: Our results suggest that metformin and quercetin have significantly (P<0.05) show a preventive effect against STZ induced diabetic and cardiac dysfunction in rats, and concomitant administration of both test drugs have shown highly significant results. Conclusion: The present study indicates the concomitant use of metformin and quercetins synergize protective effect against STZ -induced diabetic as well as cardiac dysfunctions.
Keywords: Quercetin, Metformin, cardioprotective, antioxidant, Baroreflex
Introduction
Diabetes mellitus (DM) is the most common causes of cardiovascular dysfunctions and it leads to death in patients (Scognamiglio et al., 2004). In addition, DM accelerates the increase of left ventricular dysfunction in hypertensive patients and increases cardiovascular complications (Kvan et al., 2007). Pathologic conditions, such as diabetes linked cardiomyopathy and neuropathy may lead to hyperglycemia associated oxidative stress, lipid peroxidation, metabolic disturbances and sympathetic nervous system structural and functional changes (Quan et al., 2014). All are contributors to this cardiovascular dysfunction and increasing incidence and prevalence of alterations in blood vessel structure and function, linking sensory, motor and autonomic nervous systems and depressed baroreflex sensitivity (BRS) (Dario et al., 2013;Rao et al., 2016). The proposed mechanism associated that, parasympathetic cardiac nerve dysfunction, articulated as increased resting heart rate (Rao et al., 2016).
Streptozotocin (STZ)-induced diabetes in rats causes hypotension and bradycardia, most likely related to pacemaker cell dysfunction and depression in cardiac function (Schaan et al., 2004). In fact, baroreflex-mediated bradycardia and tachycardia in response to arterial pressure (AP) alter were found to be attenuated in diabetic rats after STZ administration (Hicks et al., 1998). Changes in AP induced by STZ associated with baroreflex sensitivity, plasma renin (PRA), glucose and insulin levels and endothelium participation (Crespo et al., 2003).This impairment has been attributed parasympathetic dysfunction and alters vagal tonus has been confirmed in diabetic rats (Rao et al., 2016). Changes those are reversible by insulin and aldose reductase inhibitor treatment, suggesting a metabolic cause for this abnormality.
In this perspectives, several new approaches and a new target for treatment of diabetes; AMPK is an energy-sensing enzyme that is activated when cellular energy levels are low, and it signals to stimulate glucose uptake in skeletal muscles, fatty acid oxidation and reduces hepatic glucose production (Zhang et al., 2009). Hardie, 2008 reportrd that activation of AMPK stimulates the energy-generating pathways in liver, skeletal muscle, and heart tissues while inhibiting the energy-consuming pathways. AMPK activation results in cardioprotection by different molecular pathways that cease oxidative stress, apoptosis, and inflammatory responses, maintain vessel function and improve cardiac function in pathological cardiovascular condition (Arad et al., 2007). AMPK is a progression conserved whose activation insulin-sensitizing effects, making it an ideal therapeutic target for diabetes and its associated cardiovascular dysfunctions (da Silva et al., 2003).Now that multidrug therapy is common practice among endocrinologists; more drugs are available in a single pill. In DM early treatment initiated with combination therapy has been likely as a new approach to achieving glycemic control earlier, delay the fall of glycemic control with potential better preservation of β-cell function as well as to the management of diabetic complications (Ozra et al., 2013). However, the effects of combined treatment of these compounds on diabetes and its associated cardiovascular complications are not fully revealed. In this contest of purpose of this study to validate the potential role of the concomitant use of metformin and quercetin on diabetes associated with cardiovascular complications.
Metformin an oral Biguanide is a potent antihyperglycemic agent used as the first-line drug in the treatment of diabetes. It acts by reducing the glucose production by the liver, increasing insulin sensitivity, stimulating receptor upregulation and tissue uptake of glucose (Viollet et al., 2012). Metformin triggers AMP- protein kinase (AMPK) in hepatocytes (Hawley et al., 2002), consequently, it reduced acetyl-CoA carboxylase (ACC) activity, expression of lipogenic enzymes and it is inducing fatty acid oxidation (Zhouet al., 2001). The literature review revealed that metformin protects the heart from ischemic injury, apoptosis, inhibit inflammatory response in vascular smooth muscle cells, reduces intracellular reactive oxygen species and improves left ventricular ejection fraction in failing heart (Kravchuk et al., 2011).
In additions of antioxidant therapy also effective whether it is treat diabetes-associated cardiac complications when it is given along with diabetes treatment (Ozra et al., 2013). Bioflavonoids, a group of naturally occurring antioxidants might be used as cardioprotective agents prevent cardiotoxicity (Annapurna et al., 2009). Quercetin is the most abundant dietary flavonoid found in a wide range of fruits, vegetables, and beverages; it has potent antioxidant and anti-inflammatory properties coupled with cardioprotective action (Jeong, et al., 2012). Some investigations reported that quercetin activates AMPK and its activation implicated in the regulation of insulin action (Xu et al., 2014). A literature review revealed that quercetin shows therapeutic effects in cardiovascular disorders, atherosclerosis and retains endothelial function, ameliorates oxidative stress (Annapurna et al., 2009;Jeong, et al., 2012;Garelnabi et al., 2014).
The rationale for selection of metformin and quercetin are based on the review of literature both are drugs ameliorates oxidative stress, cardioprotective action and have anti-inflammatory property. Metformin and quercetin both have involved in activation of AMPK molecule switches that regulate energy balance of the cell. The purpose of this study is to examine the pharmacological evaluation of individual and concomitant use Quercetin and Metformin , on STZ-induced diabetes and its associated cardiovascular diseases.
Materials and methods
Drugs and chemicals
Quercetin and STZ were procured from Hi-media Mumbai and Metformin was obtained as gift sample from Swaroop Drug and Pharmaceuticals Maharashtra. Utilized various auto bioanalyzer kits to assess biochemical parameters and all the other chemicals used were of analytical grade.
Animals
Adult healthy Wistar rats (either sex) body weight 220-250g, were procured from the registered breeder. Animals were housed in appropriate polypropylene cages, under an environmentally controlled room temperature (25±1oC) and relative humidity of 45-55% under 12:12h light/dark cycle and had free access to water and rodent food. The whole experiment was carried out according to the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA) New Delhi, India and approved by the Institutional Animal Ethics Committee (IAEC) (Reg. No. ASCB/IAEC/07/14/91).
Induction of diabetes with STZ
Induction of diabetes by a single intraperitoneal (i.p.) injection of STZ (60mg/kg) (Ganda et al., 1976), dissolve in freshly prepared citrate buffer. The control group animal was injected with equal volume of vehicle. After one week of STZ administration, blood glucose level was checked by commercially available kit by auto bioanalyzer. Animals showing blood glucose level 250- 300mg/dL were considered as diabetic rats and were used in the study.
Experimental design and study protocol
Diabetic rats were randomly divided into five different groups (n=10_12) as under protocol design. Study group (C) normal vehicle control; group (STZ60) STZ administered 60mg/kg i.psingle dose; test groups (STZ60+M200) and (STZ60+Q50), STZ administered 60mg/kg i.p single dose plus metformin 200 mg/kg/ p.o. (Kravchuk et al., 2011) or quercetin 50 mg/kg/ p.o. (Annapurna et al., 2009) respectively; groups (STZ60+M200+ Q50) STZ administered 60mg/kg i.p. single dose plus metformin 200 plus quercetin 50mg/kg/ p.o. concomitantly. Daily body weight feed and water intakes were measured and Blood analysis and hemodynamic parameters evaluated after the end of the study. After the studies animals were sacrificed by decapitation to estimate isolated heart and tissue biochemical study and histopathological examination were carried out.
Measurement of hemodynamic and Baroreflex sensitivity (BRS)
Rats were anesthetized with suitable anesthesia neck was opened with a ventral midline incision and separate carotid artery cannulated with the polyethylene catheter (PE50) filled with heparinized saline (50 IU/ ml) and connected to a pressure transducer was inserted into the carotid artery. The signal was amplified by means of a bio-amplifier and monitored using the Power Lab system 4/30 (AD Instruments, NSW Australia) system using a pressure transducer for the measurements of the diastolic blood pressure (DBP), systolic blood pressure (SBP), mean arterial blood pressure (MAP) and heart rate (HR) were obtained.
After basal recording of blood pressure parameters, baroreflex sensitivity was assessed (Tauseef et al., 2007) by administering increasing doses of phenylephrine or sodium nitroprusside (20µg/ml/kg) injections and the corresponding peak reflex changes in arterial blood pressure (ABP) and heart rate (HR) was recorded after each dose of the drugs. The resultant changes in HR and corresponding rise or fall in SBP were measured at different time intervals. The relationship between the increase in SBP evoked by phenylephrine and associated tachycardia was assessed. The baroreflex sensitivity was determined as the slope of the regression line, express as bpm/mmHg was taken as an index of baroreflex sensitivity measurement. The slope obtained for each group of rats evaluated was then statistically compared.
Serum biochemical analysis
At the end of the study, the blood sample was obtained from the (overnight fasted) rats. The blood was collected then centrifuged at 2000×g for 20 min, serum was separated for analysis of different hematological parameters; glucose levels, lipid profile (TGs, T-Chol, and HDL), cardiac injury markers (CK-MB, LDH, and ALP) were quantified using commercially available kits. Meanwhile, serum insulin levels were determined by standard radioimmunoassay technique using a standard kit obtained from BRIT, BARC, Mumbai, India experiment perform in Radio Immune Assay (RIA) laboratory.
Measurement of oxidative stress level in heart tissue
Animals were sacrificed and the heart tissues were removed, washed with the cold isotonic saline and dried with filter paper. After this hearts were isolated and homogenized in 0.1 M ice-cold phosphate buffer. After centrifugation, the supernatant was used for analysis of antioxidant enzymes malondialdehyde (MDA), a measurement of Lipid peroxidation (LPO) in the heart was assayed in the form of thiobarbituric acid-reactive substances (TBARS) following the method of Ohkawa et al., 1979.Superoxide dismutase (SOD) activity level was determined according to the method of Mishra and Fridovich, 1972. Catalase (CAT) activity level was determined from the decomposition of H2O2 at 240 nm for 3 min and was monitored by using a spectrophotometer, followed the method of according to Maehly and Chance, 1954. Glutathione peroxidase levels (GPx) activity level was determined according to the method of Rotruck et al.; 1973.Nitrite is the stable end product of nitric oxide (NO) in the living system. The acquisition of nitrite was calculated in cell-free supernatants from heart homogenate by spectrophotometer assay based on Greiss reaction (Green et al., 1982).
Histology
Sacrificed animals heart was isolated, washed with ice-cold saline and fixed in 10% buffered formalin for 24h.Tissues were dehydrated in graded series of alcohol, embedded in paraffin, sectioned in 5mm thickness by using a microtome. The sections were de waxed in three changes of xylene, hydrated in two changes of 100% ethanol, followed by 95% and 80% ethanol, rinsed with water and then stained with hematoxylin and eosin (H and E). For interpretation, the pictures were taken from the prepared slides with the help of photomicroscope.
Statistical analysis
All data were represented as the mean ± SEM; comparisons in between groups were made via statistical analysis of data using one-way analysis of variance (ANOVA) compared using Newman-Keuls multiple-comparison. A p-value of <0.05 was considered as statistically significant. All statistical analyses were performed using Graph Pad Prism statistic software.
Results
Effect of individual and concomitant use of metformin and quercetin on general observations, % mortality, body weight, relative heart and left ventricular weight
The general appearance in individual groups observed like feed intake, water intake, body weight and mortality of all groups of animals was recorded throughout the experimental study. In the later days, STZ treated control group (STZ60) rats had developed clinical signs of polyuria and polydipsia, and their daily weight gain (DWG) was suppressed and some rats showed a weight loss (P<0.001) with respect compared with control saline group (C) and mortality observed in STZ control groups rats about 16%. Rest of all study groups [Test groups (STZ60+M200) and (STZ60+Q50); and concomitant treatment group (STZ60+M200+ Q50)] less remarkable changes occurred and body weight reduction have not seen remarkable reduction in comparison of (C); but in compare with (STZ60) control group, test groups and concomitant study group significantly abolish the weight reduction (P<0.001) and not seen of any mortality in duration study. Control group (STZ60) elevate relative heart and left ventricular weight respectively significant manner (P<0.001) and (P<0.01) when compared with control saline group (C), test groups (STZ60+M200) and (STZ60+Q50) significantly (P<0.05) reduced relative heart weight and concomitant treatment group (STZ60+M200+ Q50) more significantly (P<0.01) reduced relative heart weight compare with (STZ60) control group. Rest of all groups (tests and concomitant groups) was seen significant (P<0.05) relative weight reduction of left ventricular compare with (STZ60) control group (Table 1).
Table 1. Effect of individual and concomitant use of metformin and quercetin on % mortality, body weight, heart and left ventricular weight to body weight ratio
|
Parameters |
C |
STZ60 |
STZ60+M200 |
STZ60 +Q50 |
STZ60+(M200+ Q50) |
|
Mortality % |
0 |
16 |
0 |
0 |
0 |
|
Body weight (8th week) |
293.3±5.07 |
192.4±4.43a |
229.8±3.63x |
234.6±2.41x |
254.5±5.36x |
|
HW/BW (mg/g) |
3.89±0.13 |
5.84±0.17a |
4.83±0.28z |
4.66±0.97z |
4.36± 0.84y |
|
LVW/BW(mg/g) |
2.78±0.63 |
3.71±0.67b |
3.48 ±0.01 |
3.26 ±0.08z |
3.11±0.67z |
Abbreviations: HW/BW=Heart weight/Body weight ratio; LVW/BW=Left ventricular weight /Body weight ratio
Study group (C)Normal control rats injected with vehicle (citrate buffer); group (STZ 60) disease control administered (STZ 60mg/kg i.p single dose); group (STZ 60+ M 200) Metformin test group received (STZ 60mg/kg i.p single dose + Metformin 200 mg/kg/ p.o); group (STZ 60+ Q 50) Quericitine test group received (STZ 60mg/kg i.p single dose + Quercetin50 mg/kg/ p.o);group [STZ 60+ (Q 50+M 200)] both test drug combination dose groups [(STZ 60mg/kg i.p single dose + (Quercetin50 +Metformin200mg/kg/ p.o.)]. (Values expressed as mean ± SEM, n=12); aP<0.001, and bP<0.01 when compared with C; xP<0.001, yP<0.01, zP<0.05 when compared with STZ60; One way ANOVA followed by Newman-Keuls multiple-comparison post-hoc test.
Effect of individual and concomitant uses of metformin and quercetin, on fasting serum glucose, insulin and lipid levels
It is evident from the study groups treated with STZ significantly (P<0.001) increases 1st-week parameters of fasting glucose, insulin and TGs level and HDL-C level significantly (P<0.05) raised as compared with saline control (C). At the end of the study 8th-week parameters of the control group (STZ60) were significantly (P<0.001) raised fasting glucose, insulin and lipid profile as compared with saline control (C). Test groups (STZ60+M200), (STZ60+Q50) and concomitant treatment group (STZ60+M200+ Q50) 8th-week parameters fasting blood glucose level decreased significantly (P<0.01; P<0.05 and P<0.001) with respect of group when compared with control group (STZ60). Fasting insulin level 8th week in (STZ60+Q50) and (STZ60+M200+ Q50) significantly (P<0.05 and P<0.001) elevated with respect tests groups when compared with control group (STZ60). Lipid profile on 8th week parameters (TGs and T- Chol) in test groups (STZ60+Q50) significantly (P<0.05) decrease and study group (STZ60+M200+ Q50) also significantly (P<0.001 and P<0.01) reduces with respectively TGs and T- Chol parameters when compared with control group (STZ60). HDL-C level on 8th week in test groups was significantly (P<0.05) elevated in (STZ60+M200) and (STZ60+Q50) respectively and concomitant treatment group (STZ60+M200+Q50) also elevated (P<0.001) significantly when compared with control group (STZ60) (Table 2).
Table 2. Effect of individual and concomitant uses of metformin and quercetin, on fasting serum glucose, insulin and lipid levels
|
Parameters |
C |
STZ60 |
STZ60+M200 |
STZ60 +Q50 |
STZ60+ (M200+ Q50) |
|
Glucose(mg/dL) |
|||||
|
1stweek |
109.0±6.54 |
352.0±9.38a |
355.1±3.80 |
356.2±15.30 |
357.3±7.87 |
|
8thweek |
112.8±1.98 |
439.6±2.89 a |
238.0±2.20y |
281.3±7.82z |
176.6±3.32 x |
|
Insulin (μU/ml) |
|||||
|
1stweek |
15.75±2.29 |
5.72±0.43 a |
6.32±0.26 |
6.72±0.13 |
6.89±0.23 |
|
8thweek |
17.29±2.39 |
6.87±2.60 a |
8.62±5.20 |
9.57±3.80z |
12.43±2.29y |
|
LIPID PROFILE |
|||||
|
TGs (mg/dL) |
|||||
|
1stweek |
69.33±2.18 |
155.8±5.96 a |
159.8±4.60 |
160.3±6.48 |
153.6±2.04 |
|
8thweek |
73.83±3.87 |
173.7±4.61 a |
135.7±5.86 |
103.3±2.15z |
94.15±6.26x |
|
T-Chol (mg/dL) |
|||||
|
1stweek |
58.83±5.49 |
57.17±4.93 |
61.00±5.17 |
63.10±3.26 |
56.34±3.38 |
|
8thweek |
65.33±3.40 |
114.7±9.28 a |
89.33±6.71 |
86.50±5.26z |
79.23±4.91y |
|
HDL-C (mg/dL) |
|||||
|
1stweek |
46.50±3.56 |
29.06±5.54 c |
27.33±1.51 |
26.83±3.85 |
28.13±3.17 |
|
8thweek |
54.83±4.61 |
18.31± 0.64 a |
31.31± 0.64z |
35.31±1.42z |
43.62±3.12x |
Abbreviations: TGs=Triglycerides; T-Chol=Total cholesterol; HDL-C= High-density lipoprotein cholesterol.
Study group (C)Normal control rats injected with vehicle (citrate buffer); group (STZ 60) disease control administered (STZ 60mg/kg i.p single dose); group (STZ 60+ M 200) Metformin test group received (STZ 60mg/kg i.p single dose + Metformin 200 mg/kg/ p.o); group (STZ 60+ Q 50) Quericitine test group received (STZ 60mg/kg i.p. single dose + Quercetin50 mg/kg/ p.o.);group [STZ 60+ (Q 50+M 200)] both test drug combination dose groups [(STZ 60mg/kg i.p single dose + (Quercetin50 +Metformin200mg/kg/ p.o.)]. (Values expressed as mean ± SEM, n=12); aP<0.001, and cP<0.05 when compared with C; xP<0.001,yP<0.01,zP<0.05 when compared with STZ60; One way ANOVA followed by Newman-Keuls multiple-comparison post-hoc test.
Effect of individual and concomitant use of metformin and quercetin, on serum cardiac injury markers and oxidative stress level in the heart
STZ treated control group (STZ60) elevated significantly (P<0.01) serum cardiac injury markers CK-MB level and also elevated LDH and ALP levels significantly (P<0.001) when compared with control group (C). Test groups (STZ60+M200), (STZ60+Q50) and concomitant treatment group (STZ60+M200+ Q50) reduced cardiac injury markers CK-MB level (P<0.05) with respect of the study groups when compared with control group (STZ60). In similar cardiac injury marker, LDH reduced significantly (P<0.05) in tests groups (STZ60+M200), (STZ60+Q50) and in the concomitant treatment group also reduced significantly (P<0.01) when compared with control group (STZ60). ALP level reduced significantly (P<0.05) in test groups (STZ60+M200), in the test group (STZ60+Q50) and in the concomitant treatment group (STZ60+M200+ Q50) also reduced ALP level significantly (P<0.01) when compared with control group (STZ60).
STZ treated control group (STZ60) elevate LPO and nitrites level significantly (P<0.01) and decrease SOD, CAT and GPx levels significantly (P<0.001) when compared with control group (C). In tests groups (STZ60+M200), (STZ60+Q50) and in concomitant treatment group (STZ60+M200+ Q50) reduced LPO level significantly (P<0.05; P<0.01and P<0.001) with respect of the group and decrease nitrites level tests groups (STZ60+M200) significantly (P<0.05) and in (STZ60+Q50) and (STZ60+M200+ Q50) groups reduced significantly (P<0.01) when compared with control group (STZ60). SOD, CAT and GPx levels in tests groups (STZ60+M200) significantly (P<0.05) elevate, in similar parameters in tests groups (STZ60+Q50) significantly (P<0.05) maintain normal SOD, CAT level and and GPx level significantly (P<0.01) maintain normal, in concomitant treatment group (STZ60+M200+ Q50) elevate significantly (P<0.01) SOD, CAT and GPx level significantly (P<0.001) maintain normal when compared with control group (STZ60) (Table 3).
Table 3. Effect of individual and concomitant use of metformin and quercetin, on serum cardiac injury marker, andoxidative stress level in the heart
|
Parameters |
C |
STZ60 |
STZ60+M200 |
STZ60 +Q50 |
STZ60+ (M200+ Q50) |
|
Serum cardiac injury markers |
|||||
|
CK-MB(IU/L) |
65.07±2.31 |
96.07±0.46b |
77.14±7.22z |
75.78±1.08 z |
68.84±5.15 z |
|
LDH(IU/L) |
778.5 ± 5.1 |
970.5±6.8a |
874.5±4.1z |
853.9±3.3 z |
784.0±4.24 y |
|
ALP(IU/L) |
125.8±6.3 |
246.6±5.36a |
161.43±1.4z |
172.8±2.17y |
132.5±6.34y |
|
Oxidative stress level in heart |
|||||
|
LPO (nM MDA/mg of protein) |
1.77±1.43 |
23.54±6.64 a |
12.91±0.64z |
9.73±8.91y |
4.61±6.12x |
|
SOD (U/mg of protein) |
9.15±0.43 |
4.11±8.12 b |
6.18±0.65z |
6.53±4.26z |
7.74±9.15y |
|
CAT (nM/mg of protein/min) |
4.63±8.12 |
1.16±4.21 b |
2.91±8.87z |
2.83±2.10z |
3.87±0.43y |
|
GPx (nM/mg of protein/min) |
21.26±3.62 |
4.81±7. 82 a |
8.72±5.41z |
15.15±3.47y |
17.64±7.43x |
|
Nitrites level (μM nitrite/ml) |
15.31±1.45 |
52.34± 5.21a |
31.64± 4.62z |
21.83±4.10y |
16.52±3.22y |
Abbreviations: CK-MB = Creatinekinaseisoenzyme; LDH = Lactate dehydrogenase; ALP=Alkaline Phosphatase; LPO = Lipid peroxidation, SOD = Superoxide dismutase, CAT = catalase and GPx = gluthathione peroxidase levels.
Study group (C)Normal control rats injected with vehicle (citrate buffer); group (STZ 60) disease control administered (STZ 60mg/kg i.p. single dose); group (STZ 60+ M 200) Metformin test group received (STZ 60mg/kg i.p single dose + Metformin 200 mg/kg/ p.o.); group (STZ 60+ Q 50) Quericitine test group received (STZ 60mg/kg i.p. single dose + Quercetin50 mg/kg/ p.o.);group [STZ 60+ (Q 50+M 200)] both test drug combination dose groups [(STZ 60mg/kg i.p single dose + (Quercetin50 +Metformin200mg/kg/ p.o.)]. (Values expressed as mean ± SEM, n=12).aP<0.001, and bP<0.01, when compared with C; xP<0.001,yP<0.01,zP<0.05 when compared with STZ 60; One way ANOVA followed by Newman-Keuls multiple-comparison post-hoc test.
Effect of individual and concomitant use of metformin and quercetin on hemodynamic and baroreflex sensitivity
STZ treated control group (STZ60) significantly (P<0.05) reduced DBP, SBP, and MAP and also reduced HR significantly (P<0.01), baroreflex sensitivity (phenylephrine and sodium nitroprusside) significantly (P<0.001) reduced when compared with control group (STZ60). Tests groups (STZ60+M200) significantly (P<0.05) maintain near to normal DBP, SBP, and HR as well as baroreflex sensitivity significantly (P<0.01) elevated in a similar group when compared with the control group (STZ60). In tests groups (STZ60+Q50) significantly (P<0.05) maintain normal DBP, SBP, MAP, and HR as well as baroreflex sensitivity significantly (P<0.001) maintain near to normal in the study group when compare with the control group (STZ60).Concomitant treatment group (STZ60+M200+ Q50) significantly (P<0.01) maintain normal DBP, SBP and significantly (P<0.05; P<0.001) maintain MAP and HR as well as baroreflex sensitivity significantly (P<0.001) maintain near to normal in the study group when compare with the control group (STZ60), (Table 4).
Table 4. Effect of individual and concomitant use of metformin and quercetin on parameters of basal hemodynamic and baroreflex sensitivity
|
Parameters |
C |
STZ60 |
STZ60+M200 |
STZ60 +Q50 |
STZ60+ (M200+ Q50) |
|
|
Basal Hemodynamic parameters |
|
|||||
|
DBP(mmHg) |
95.24±0.97 |
83.6±1.21c |
87.71±2.33z |
89.94±1.71z |
91.23±4.13y |
|
|
SBP (mmHg) |
126.6±3.04 |
113.8±1.10c |
117.2±3.13z |
118.5±2.62z |
121.7±3.71y |
|
|
MAP (mmHg) |
107.9±0.82 |
94.7±0.69c |
96.0±0.98 |
99.4±1.14z |
101.8±6.44z |
|
|
HR(BPM) |
337.5±1.70 |
279.1±1.93b |
289.3±3.76z |
296.3±0.56z |
309.7±6.24x |
|
|
Baroreflex sensitivity(Beats/min/mmHg) |
||||||
|
Phenylephrine |
2.23±4.64 |
0.43±3.06a |
0.91±5.26y |
1.82±1.31x |
1.93±7.27x |
|
|
Sodium nitroprusside |
0.86±3.92 |
0.18±9.32a |
0.43±1.93y |
0.65±2.04x |
0.79±2.92x |
|
Abbreviations: DBP = Daiastoloc blood pressur; SBP = Systolic blood pressure; MAP = Mean arteliarprsessur; HR = Heart Rate
Study group (C)Normal control rats injected with vehicle (citrate buffer); group (STZ 60) disease control administered (STZ 60mg/kg i.p single dose); group (STZ 60+ M 200) Metformin test group received (STZ 60mg/kg i.p. single dose + Metformin 200 mg/kg/ p.o.); group (STZ 60+ Q 50) Quericitine test group received (STZ 60mg/kg i.p. single dose + Quercetin50 mg/kg/ p.o.);group [STZ 60+ (Q 50+M 200)] both test drug combination dose groups [(STZ 60mg/kg i.p. single dose + (Quercetin50 +Metformin200mg/kg/ p.o.)]. (Values expressed as mean ± SEM, n=12)aP<0.001;bP<0.01, and cP<0.05, when compared with C; xP<0.001,yP<0.01,zP<0.05 when compared with STZ 60; One way ANOVA followed by Newman-Keuls multiple-comparison post-hoc test.
Effect of individual and concomitant use of metformin and quercetin on heart histopathological changes
STZ treated control group (STZ60) shows an increase in infarct area, necrosis, and separation of myocardial fibers. In tests groups (STZ60+M200), (STZ60+Q50) show a mild change in the infarct area, necrosis, and separation of myocardial fibers. Concomitant treatment group (STZ60+M200+ Q50) showed minute infract area, necrosis, and separation of myocardial fibers when compared with control group (STZ60) (Figure.1 and Table.5).
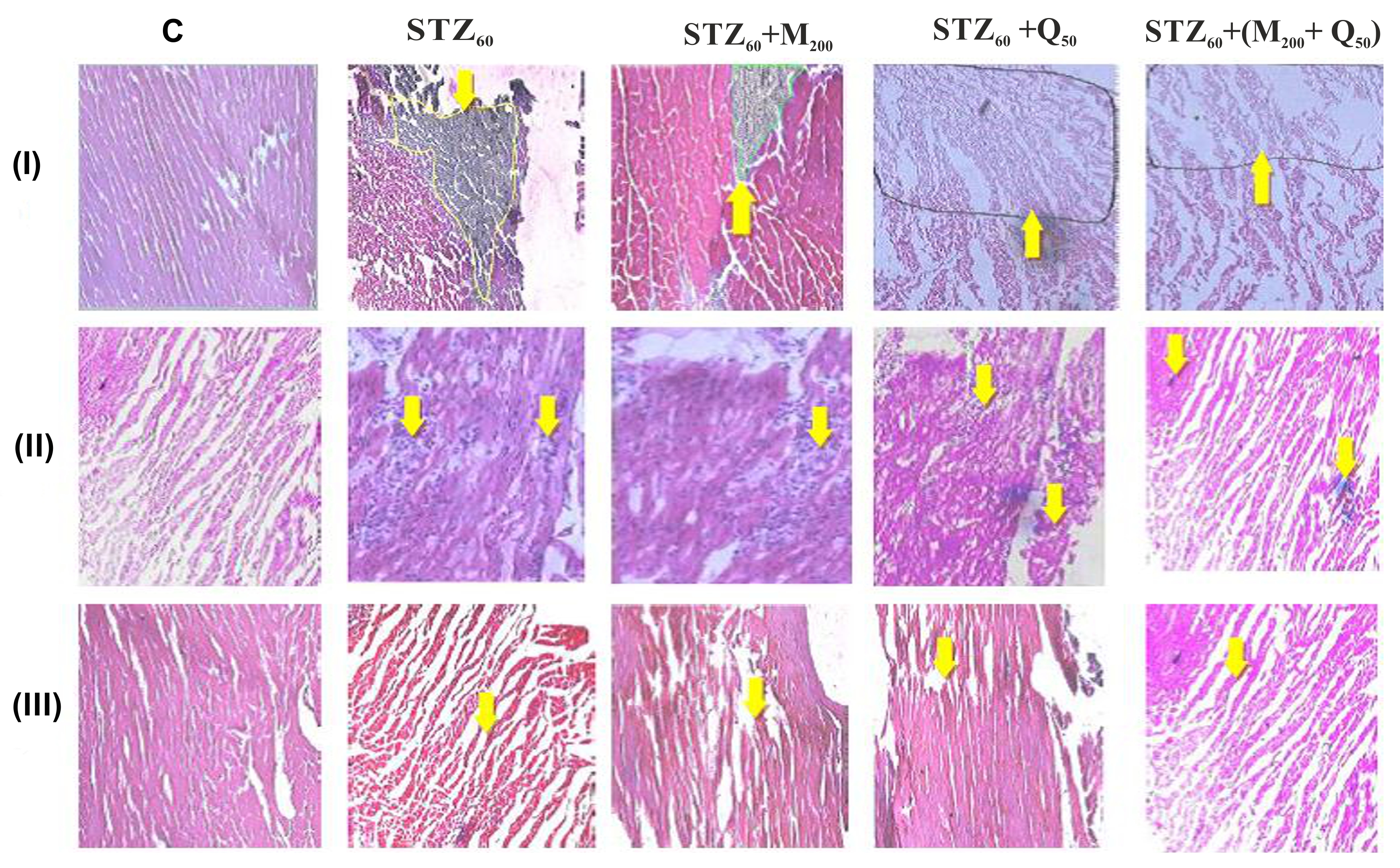
Figure 1. Effect of individual and concomitant use of metformin and quercetin on histopathological changes in the rat heart tissues.
Figure 1 (I, II and III) Represent images of histopathological examinations of heart- infarct area, Necrosis and separation of myocardial fibers respectively; Study group (C) Normal control rats injected with vehicle (citrate buffer); group (STZ 60) disease control administered (STZ 60mg/kg i.p single dose);; group (STZ 60+ M 200) Metformin test group received (STZ 60mg/kg i.p single dose + Metformin 200 mg/kg/ p.o); group (STZ 60+ Q 50) Quericitine test group received (STZ 60mg/kg i.p single dose + Quercetin50 mg/kg/ p.o) group [STZ 60+ (Q 50+M 200)] both test drug combination dose groups [(STZ 60mg/kg i.p single dose + (Quercetin50 +Metformin200mg/kg/ p.o.)].Heart sections were fixed and stained with hematoxylin and eosin (H&E) stain. Images were taken at 100× magnification and scale bar = 10µM. In Figure 1 (I, II and III) group (C) showed clear normal integrity of myocardial cell membrane; in group (STZ 60) examination of the showed marked myonecrosis, edema, muscle separation and infiltration of inflammatory cells were observed (arrow).The reduction of histopathological scores in (STZ 60+M200) and (STZ 60+Q50)also showed less edema and necrotic (arrow) changes and group [STZ 60+ (Q 50+M 200)] showed, remarkably histological protection of the myocardium.
Table 5. Effect of individual and concomitant use of metformin and quercetin on heart histopathological changes (Severity Index).
|
Parameters |
C |
STZ60 |
STZ60+M200 |
STZ60 +Q50 |
STZ60+ (M200+ Q50) |
|
Infarct Area |
0 |
4 |
3 |
2 |
1 |
|
Necrosis |
0 |
3 |
2 |
1 |
1 |
|
Separation of myocardial fibres |
0 |
4 |
2 |
2 |
1 |
Study group (C)Normal control rats injected with vehicle (citrate buffer); group (STZ 60) disease control administered (STZ 60mg/kg i.p single dose); group (STZ 60+ M 200) Metformin test group received (STZ 60mg/kg i.p. single dose + Metformin 200 mg/kg/ p.o.); group (STZ 60+ Q 50) Quericitine test group received (STZ 60mg/kg i.p. single dose + Quercetin50 mg/kg/ p.o.;group [STZ 60+ (Q 50+M 200)] both test drug combination dose groups [(STZ 60mg/kg i.p. single dose + (Quercetin50 +Metformin200mg/kg/ p.o.)].
Discussion
Diabetes mellitus increases the risk of heart failure independent of underlying coronary artery disease and may lead to cardiomyopathy (Kvan et al., 2007). Diabetic cardiomyopathy on (DCM) either on animal or molecular study, have found that myocardial abnormal glucose utilization and the shift toward fatty acid oxidation are the major pathophysiological alterations, that may lead to diabetes mellitus (DM)-associated myocardial remodeling and heart failure (Quanet al., 2014; Dario et al., 2013).
STZ induced toxicity has been well documented in experimental animals, administration of single i.p. a dose of STZ induced diabetes it’s led to hyperglycemia and oxidative stress associated cardiotoxicity (Ganda et al., 1976). STZ induced diabetes brings changes in endothelial damage and mitochondrial membrane leads to cardiac dysfunction (Hong et al., 2005). From a previous experimental study reported that STZ treated rats had developed clinical signs of polyuria and polydipsia during the experimental (Ganda et al., 1976). In present study single i.p. a dose of STZ (60 mg/kg) display marked changes in rats had developed clinical signs such as polyuria and polydipsia, and their daily weight gain (DWG) was suppressed and some rats showed a weight loss, mortality (16%), increase in heart weight and the decrease in body weight. The causes of decrease in the body weight may be due to some metabolic changes and increase the heart weight might be attributed to increased edema, vascular hemorrhage or extensive necrosis of cardiac muscle fibers followed by invasion of damaged tissues with inflammatory cells (Crespo et al., 2003;Ganda,1980). In the present study, STZ control group significantly increased relative heart and left ventricle weight and decreased body weight respect of control group. These changes were less prominent in metformin, quercetin and concomitant treated group and whereas in these groups was seen no mortality, decrease in relative heart and left ventricle weight and maintain near to normal body weight with respect of STZ treated control group. To abolish of STZ toxicity sine by metformin and quercetin is due to its ability to ameliorate oxidative stress, both play a major role in improving activity and level of serum biomarker and anti-inflammatory nature.
There is a strong relationship between hyperglycemia and oxidative stress in diabetic cardiomyopathy (Dario et al., 2013). Hyperglycemia marks in a series of events leading to lowered total antioxidant status and hence progressive functional and structural alterations of cardiovascular function in diabetes mellitus (Klinsmann et al., 2014).In the present study, hyperglycemia-induced oxidative stress was established by increased lipid peroxidation, increased generation of superoxide anions and reduced glutathione levels in the diabetic STZ control group. Recent findings have described that the hypoglycemic effect of metformin occurs through activation of AMPK, a serine/ threonine protein kinase a central role in the regulation of cellular metabolism and energy balance (Hawley et al., 2002). In addition, some natural compounds (polyphenol) activate AMPK during its beneficial effect on metabolic disorders (Zang et al., 2006).Therefore, it can be suggested that polyphenol are potential compounds of natural origin to diabetes treatment because they stimulate a cellular target similar to that of metformin (Annapurna et al., 2009; Xu et al., 2014). In a similar way observed with quercetin and metformin, showing that the antihyperglycemic effect could be attributed, to AMPK stimulation.
The experimental study shows biochemical changes as well as oxidative damage in the cardiac tissue after treatment with STZ, its ability to destruct myocardial cells leading to direct myocardial endothelial damage. As a result, CK-MB, LDH, and ALP are released into the bloodstream; elevated levels of these enzymes are associated with certain types of heart damage such as myocardial infarction, myocarditis and heart failure (Walker, 2006). In the present study i.p. administered STZ group showed the significant elevation in the levels of CK-MB, LDH, and ALP when compared with control. Administration of metformin and quercetin and in combination results significantly reduced in the elevated level of CK-MB, LDH, and ALP when compared to STZ control. This reduction in the enzyme level confirms that both metformin and quercetin either alone or in combination is responsible for maintenance of normal structural and architectural integrity of cardiac myocytes, thereby restricting the leakage of these enzymes, which can be accounted for its membrane stabilizing property, it is also confirmed in histopathological examination.
STZ administration is associated with a decrease in endogenous antioxidants and increase in oxygen free radicals resulting in increased oxidative stress (Ganda et al., 1976), which is followed by the development of a variety of subcellular changes in the myocardium. Oxidative stress has been strongly associated with tissue damage in diabetic individuals (Dario et al., 2013; Klinsmann et al., 2014). In the present study, the diabetic control group showed a significant elevation in the level of LPO and Nitrite level. Reduction in the antioxidants levels, strong evidence of increased oxidative stress and its lead to tissue damage (Klinsmann et al., 2014).Oral administration of metformin and quercetin alone or in combination significantly has been reported to possess free radical scavenger activity and able to activate cellular antioxidative enzyme levels and will be stored the normal cell structure in the heart.
Hyperglycemic rat causes hypotension and bradycardia, most likely related to pacemaker cell dysfunction and depression in cardiac function, myocardial fibrosis, and left ventricular hypertrophy, systolic and diastolic dysfunction (Schaan et al., 2004; Hicks et al., 1998). In the present study administration of STZ showed the significant decrease in DBP, SBP, MAP, and HR when compared with control. In fact, baroreflex-mediated bradycardia and tachycardia in response to arterial pressure (AP) alter were found to be attenuated in diabetic rats (Hicks et al., 1998). This impairment has been attributed, partially or fully parasympathetic dysfunction and alters vagal tonus (Rao et al., 2016). The previous study reported that changes that are reversible by insulin and aldose reductase inhibitor treatment, suggesting a metabolic cause for this abnormality (Crespo et al., 2003). In the similar oral administration of metformin and quercetin alone or in combination results substantial maintain near to normal hemodynamic and baroreflex sensitivity parameters. It is strongly supported that both treatments revert insulin sensitivity and improve metabolism.
In normal rats, cardiac muscle fibers were found to be of uniform size, shape, and configurations with no inflammatory cell infiltrate. Cardiotoxicity showing a varying degree of vacuolar changes in the cardiac muscle fibers, interstitial edema, infiltration of inflammatory intravascular thrombus and myofibril loss (Crespo et al., 2003; Ganda,1980). STZ treated hyperglycemic rats produced all massive change in the myocardium. In present study treatment with metformin and either alone or in combination inhibits hyperglycemia-induced cardiac damage by reversal of infiltration of inflammatory cells and fragmentation of myofibrils and contraction band necrosis.
Conclusion
It is summarized that overall cardioprotective effect of metformin and quercetin is due to its ability to ameliorate oxidative stress; both play a major role in improving activity and level of serum cardiac injury biomarker and normalized heart rate. Anti-inflammatory nature of both metformin and quercetin plays important role in the reversal of inflammation markers observed during histopathological and biochemical estimation findings. In treatment groups, metformin and quercetin comparable effective conclude from biochemical, hemodynamic, and histopathological findings. In conclusion, our results suggest that metformin, quercetin, and especially their cotreatment have the potential to improve diabetes and cardiotoxic sins in STZ-induced diabetic rats. On the basis these findings, it may be worthy to suggest that concomitant administration of metformin and quercetin produce synergistic effect along with improves cardiac function.
Acknowledgements
The authors would like to thanks to the management committee of ASBASJS Memorial College of Pharmacy BELA (Ropar) and IK Gujral Punjab Technical University, Jalandhar, Kapurthala, Punjab, India, for providing facilities and registration to conduct the project work.
Competing of interest statement
Authors’ have declared that we have no competing or conflict of interest exists.
References
Annapurna A, Reddy CS, Akondi RB, Rao SR. 2009.Cardioprotective actions of two bioflavonoids, quercetin and rutin, in experimental myocardial infarction in both normal and streptozotocin-induced type I diabetic rats. Journal of Pharmacy and Pharmacology, 61(10):1365–1374.
Arad M, Seidman CE, Seidman JG. 2007. AMP-activated protein kinase in the heart: Role during health and disease. Circulation Research, 100(4):474-488.
Crespo MJ, Moreta S, Gonzalez J. 2003.Cardiovascular Deterioration in STZ-Diabetic Rats: Possible Role of Vascular RAS. Pharmacology, 68:1–8.
da Silva XG, Leclerc I, Varadi A, Tsuboi T, Moule SK, Rutter GA. 2003. Role for AMP-activated protein kinase in glucose-stimulated insulin secretion and preproinsulin gene expression. Biochemical Journal, 371(3):761–774.
Dario P, Manfredi T, Rizzi A, Giovanni G, Carmine C. 2013. Oxidative Stress in Diabetes: Implications for Vascular and Other Complications. International Journal of Molecular Sciences, 14(11):21525–21550.
Ganda OP, Rossini AA, Like AA.1976. Studies on streptozotocin diabetes. Diabetes, 25(7):595-603.
Ganda OP. 1980. Pathogenesis of macrovasculardiseasein the human diabetic. Diabetes, 29:931–942.
Garelnabi M, Mahini H, Wilson T. 2014. Quercetin intake with exercise modulates Lipoprotein metabolism and reduces atherosclerosis plaque formation. Journal of International Society of Sports Nutrition, 11:22.
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR.1982. Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Analytical Biochemistry, 126(1):131-138.
Hardie DG. 2008. AMPK: A key regulator of energy balance in the single cell and the whole organism. International Journal of Obesity, 32(9):S7-S12.
Hawley SA, Gadalla AE, Olsen GS,Hardie DG.2002. The Antidiabetic Drug Metformin Activates the AMP-Activated Protein Kinase Cascade via an Adenine Nucleotide-Independent Mechanism. Diabetes, 51(8):2420-2425.
Hicks KK, Seifen E, Stimers JR, Kennedy RH. 1998. Effects of streptozotocin-induced diabetes on heart rate, blood pressure and cardiac autonomic nervous control. Journal of the Autonomic Nervous System, 69(1):21-30.
Hong D, Michael H, William BW, Winnie L, Jacqueline M, Julianne R, Chris T. 2005. Endothelial dysfunction in the streptozotocin-induced diabetic apoE-deficient mouse.British Journal of Pharmacology,146(8):1110–1118.
Jeong SM, Kang MJ, Choi HN, Kim JH, Kim JI. 2012. Quercetin ameliorates hyperglycemia and dyslipidemia and improves antioxidant status in type 2 diabetic db/db mice. Nutrition Research and Practice, 6(3):201–207.
Klinsmann CS, Camila PB, Pedro OB, Fabio RF, Ary FJ, Ana AH. 2014. Cardiac Energy Metabolism and Oxidative Stress Biomarkers in Diabetic Rat Treated with Resveratrol. PLoS ONE, 9(7):1-8.
Kravchuk E, Grineva E, Bairamov A, Galagudza M, Vlasov T. 2011.The effect of Metformin on the myocardial tolerance to ischemia-reperfusion injury in the rat model of diabetes mellitus Type II. Experimental Diabetes Research, 10:1-5.
Kvan E, Pettersen KI, Sandvik L, Reikvam A. 2007. High mortality in diabetic patients with acute myocardial infarction: cardiovascular co-morbidities contribute most to the high risk. International Journal of Cardiology, 121 (2):184–188.
Maehly AC, Chance B, 1954. The assay of catalases and peroxidases. Methods of Biochemical Analysis, 1:357–424.
Misra HP, Fridovich I. 1972. The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. The Journal of Biological Chemistry, 247(10):3170–3175.
Ohkawa H, Ohishi N, Yagi K. 1979. Assays of lipid peroxides in animal tissues by thiobarbituric acid reaction. Analytical Biochemistry, 95:351– 358.
Ozra TM, Bagher L, Mohammad A. 2013. A novel management of diabetes by men of strong antioxidants’ combination. Journal of Medical Hypothesis and Ideas, 7(1):25-30.
Quan L, Shudong W, Lu C. 2014. Diabetic cardiomyopathy and its mechanisms: Role of oxidative stress and damage. Journal of Diabetes Investigation, 5(6):623–634.
RaoAD, Bonyhay I, DankwaJ, Baimas GM, KneenL, BallatoriS, Freeman R, Adler GK. 2016. Baroreflex Sensitivity Impairment during Hypoglycemia: Implications for Cardiovascular Control. Diabetes. 65(1):209–215.
Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoekstra WG. 1973. Selenium: biochemical role as a component of glutathione peroxidase. Science, 179:588–590.
Schaan BD, DallAg P, Maeda CY, Ferlin E, Fernandes TG, Schmid H, Irigoyen MC. 2004. Relationship between cardiovascular dysfunction and hyperglycemia in streptozotocin-induced diabetes in rats. Brazilian Journal of Medical and Biological Research, 37(12):1895-902.
Scognamiglio R, Avogaro A, Negut C, Piccolotto R, Vigili de KS, Tiengo A. 2004. Early myocardial dysfunction in the diabetic heart: current research and clinical applications. American Journal of Cardiology, 93(8A):17A–20A.
Tauseef M, Sharma KK, Fahim M. 2007. Aspirin restores normal baroreflex function in hypercholesterolemic rats by its antioxidative action. European Journal of Pharmacology, 556(1-3):136-143.
Viollet B, Guigas B, Sanz Garcia N, Leclerc J, Foretz M, Andreelli F. 2012. Cellular and molecular mechanisms of metformin: an overview. Clinical Science, 122(6):253-270.
Walker DB. 2006. Serum Chemical Biomarkers of Cardiac Injury for Nonclinical Safety Testing. Toxicologic Pathology, 34:94–104.
Xu M, Hu J, Zhao W, Gao X, Jiang C, Liu K, Liu B, Huang F. 2014. Quercetin differently regulates insulin-mediated glucose transporter 4 translocation under basal and inflammatory conditions in adipocytes. Molecular Nutrition and Food Research, 58(5):931-941.
Zang M, Xu S, Maitland-Toolan KA, Zuccollo A, Hou X, Jiang B, Wierzbicki M, Verbeuren TJ, Cohen RA. 2006. Polyphenols stimulate AMP-activated protein kinase, lower lipids, and inhibit accelerated atherosclerosis in diabetic LDL receptor-deficient mice. Diabetes, 55(8):2180–2191.
Zhang BB, Zhou G, Li C. 2009. AMPK: An Emerging Drug Target for Diabetes and the Metabolic Syndrome. Cell Metabolism, 9(5):407-416.
Zhou G, Myers R, Li Y, Chen Y, ShenX, Melody JF, John MW,Ventre,DoebberT, FujiiN, MusiN, Hirshman MF, Goodyear JL ,Moller ED. 2001. Role of AMP-activated protein kinase in mechanism of metformin action. The Journal of Clinical Investigation, 108(8):1167–1174.